Endothelial progenitor cell: a blood cell by many other names may serve similar functions
- PMID: 23371317
- PMCID: PMC3704045
- DOI: 10.1007/s00109-013-1002-8
Endothelial progenitor cell: a blood cell by many other names may serve similar functions
Abstract
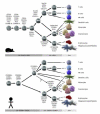
The first reports of circulating cells that displayed the capacity to repair and regenerate damaged vascular endothelial cells as progenitor cells for the endothelial lineage (EPC) were met with great enthusiasm. However, the cell surface antigens and colony assays used to identify the putative EPC were soon found to overlap with those of the hematopoietic lineage. Over the past decade, it has become clear that specific hematopoietic subsets play important roles in vascular repair and regeneration. This review will provide some overview of the hematopoietic hierarchy and methods to segregate distinct subsets that may provide clarity in identifying the proangiogenic hematopoietic cells. This review will not discuss those circulating viable endothelial cells that play a role as EPC and are called endothelia colony-forming cells. The review will conclude with identification of some roadblocks to progress in the field of identification of circulating cells that participate in vascular repair and regeneration.
Figures

Similar articles
-
Endothelial progenitor cells--potential new avenues to improve neoangiogenesis and reendothelialization.Int Rev Cell Mol Biol. 2013;306:43-81. doi: 10.1016/B978-0-12-407694-5.00002-X. Int Rev Cell Mol Biol. 2013. PMID: 24016523 Review.
-
Methodological development of a clonogenic assay to determine endothelial progenitor cell potential.Circ Res. 2011 Jun 24;109(1):20-37. doi: 10.1161/CIRCRESAHA.110.231837. Epub 2011 May 12. Circ Res. 2011. PMID: 21566217
-
Defining human endothelial progenitor cells.J Thromb Haemost. 2009 Jul;7 Suppl 1:49-52. doi: 10.1111/j.1538-7836.2009.03407.x. J Thromb Haemost. 2009. PMID: 19630767 Review.
-
Unresolved questions, changing definitions, and novel paradigms for defining endothelial progenitor cells.Blood. 2005 Sep 1;106(5):1525-31. doi: 10.1182/blood-2005-04-1509. Epub 2005 May 19. Blood. 2005. PMID: 15905185 Review.
-
Biology and flow cytometry of proangiogenic hematopoietic progenitors cells.Cytometry A. 2015 Jan;87(1):5-19. doi: 10.1002/cyto.a.22596. Epub 2014 Nov 21. Cytometry A. 2015. PMID: 25418030 Free PMC article. Review.
Cited by
-
Endothelial angiogenesis is directed by RUNX1T1-regulated VEGFA, BMP4 and TGF-β2 expression.PLoS One. 2017 Jun 22;12(6):e0179758. doi: 10.1371/journal.pone.0179758. eCollection 2017. PLoS One. 2017. PMID: 28640846 Free PMC article.
-
Targeting Angiotensin-Converting Enzyme-2/Angiotensin-(1-7)/Mas Receptor Axis in the Vascular Progenitor Cells for Cardiovascular Diseases.Mol Pharmacol. 2021 Jan;99(1):29-38. doi: 10.1124/mol.119.117580. Epub 2020 Apr 22. Mol Pharmacol. 2021. PMID: 32321734 Free PMC article. Review.
-
Atherosclerotic Pre-Conditioning Affects the Paracrine Role of Circulating Angiogenic Cells Ex-Vivo.Int J Mol Sci. 2020 Jul 24;21(15):5256. doi: 10.3390/ijms21155256. Int J Mol Sci. 2020. PMID: 32722151 Free PMC article.
-
Vitamin D Status in Rheumatoid Arthritis: Inflammation, Arterial Stiffness and Circulating Progenitor Cell Number.PLoS One. 2015 Aug 4;10(8):e0134602. doi: 10.1371/journal.pone.0134602. eCollection 2015. PLoS One. 2015. PMID: 26241902 Free PMC article.
-
Therapeutic Potential of Endothelial Colony-Forming Cells in Ischemic Disease: Strategies to Improve their Regenerative Efficacy.Int J Mol Sci. 2020 Oct 7;21(19):7406. doi: 10.3390/ijms21197406. Int J Mol Sci. 2020. PMID: 33036489 Free PMC article. Review.
References
-
- Chavakis E, Koyanagi M, Dimmeler S. Enhancing the outcome of cell therapy for cardiac repair: progress from bench to bedside and back. Circulation. 2010;121:325–335. - PubMed
-
- Losordo DW, Dimmeler S. Therapeutic angiogenesis and vasculogenesis for ischemic disease: part II: cell-based therapies. Circulation. 2004;109:2692–2697. - PubMed
-
- Kang S, Yang YJ, Li CJ, Gao RL. Effects of intracoronary autologous bone marrow cells on left ventricular function in acute myocardial infarction: a systematic review and meta-analysis for randomized controlled trials. Coron Artery Dis. 2008;19:327–335. - PubMed
-
- Bai Y, Sun T, Ye P. Age, gender and diabetic status are associated with effects of bone marrow cell therapy on recovery of left ventricular function after acute myocardial infarction: a systematic review and meta-analysis. Ageing Res Rev. 2010;9:418–423. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

