Functional mapping of the neuronal substrates for drug tolerance in Drosophila
- PMID: 23371357
- PMCID: PMC4586076
- DOI: 10.1007/s10519-013-9583-0
Functional mapping of the neuronal substrates for drug tolerance in Drosophila
Abstract
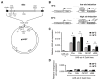
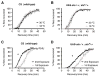
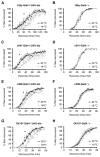
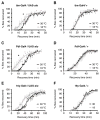
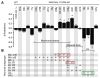
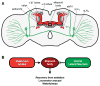
Physical dependence on alcohol and anesthetics stems from neuroadaptive changes that act to counter the effects of sedation in the brain. In Drosophila, exposure to either alcohol or solvent anesthetics have been shown to induce changes in expression of the BK-type Ca(2+)-activated K(+) channel gene slo. An increase in slo expression produces an adaptive modulation of neural activity that generates resistance to sedation and promotes drug tolerance and dependence. Increased BK channel activity counteracts the sedative effects of these drugs by reducing the neuronal refractory period and enhancing the capacity of neurons for repetitive firing. However, the brain regions or neuronal populations capable of producing inducible resistance or tolerance remain unknown. Here we map the neuronal substrates relevant for the slo-dependent modulation of drug sensitivity. Using spatially-controlled induction of slo expression we identify the mushroom bodies, the ellipsoid body and a subset of the circadian clock neurons as pivotal regions for the control of recovery from sedation.
Figures
Similar articles
-
A histone modification identifies a DNA element controlling slo BK channel gene expression in muscle.J Neurogenet. 2015;29(2-3):124-34. doi: 10.3109/01677063.2015.1050097. Epub 2015 Jul 13. J Neurogenet. 2015. PMID: 25967280 Free PMC article.
-
BK channels play a counter-adaptive role in drug tolerance and dependence.Proc Natl Acad Sci U S A. 2010 Sep 14;107(37):16360-5. doi: 10.1073/pnas.1005439107. Epub 2010 Aug 26. Proc Natl Acad Sci U S A. 2010. PMID: 20798347 Free PMC article.
-
Homeostatic control of neural activity: a Drosophila model for drug tolerance and dependence.Int Rev Neurobiol. 2011;99:23-50. doi: 10.1016/B978-0-12-387003-2.00002-1. Int Rev Neurobiol. 2011. PMID: 21906535 Free PMC article. Review.
-
A DNA element in the slo gene modulates ethanol tolerance.Alcohol. 2016 Mar;51:37-42. doi: 10.1016/j.alcohol.2015.12.003. Epub 2016 Jan 21. Alcohol. 2016. PMID: 26992698 Free PMC article.
-
Ethanol interactions with calcium-dependent potassium channels.Alcohol Clin Exp Res. 2007 Oct;31(10):1625-32. doi: 10.1111/j.1530-0277.2007.00469.x. Alcohol Clin Exp Res. 2007. PMID: 17850640 Review.
Cited by
-
The foraging gene affects alcohol sensitivity, metabolism and memory in Drosophila.J Neurogenet. 2021 Sep;35(3):236-248. doi: 10.1080/01677063.2021.1931178. Epub 2021 Jun 7. J Neurogenet. 2021. PMID: 34092172 Free PMC article.
-
Normal Ethanol Sensitivity and Rapid Tolerance Require the G Protein Receptor Kinase 2 in Ellipsoid Body Neurons in Drosophila.Alcohol Clin Exp Res. 2020 Aug;44(8):1686-1699. doi: 10.1111/acer.14396. Epub 2020 Jul 9. Alcohol Clin Exp Res. 2020. PMID: 32573992 Free PMC article.
-
Flying Together: Drosophila as a Tool to Understand the Genetics of Human Alcoholism.Int J Mol Sci. 2020 Sep 11;21(18):6649. doi: 10.3390/ijms21186649. Int J Mol Sci. 2020. PMID: 32932795 Free PMC article. Review.
-
Excitatory and inhibitory neural dynamics jointly tune motion detection.Curr Biol. 2022 Sep 12;32(17):3659-3675.e8. doi: 10.1016/j.cub.2022.06.075. Epub 2022 Jul 21. Curr Biol. 2022. PMID: 35868321 Free PMC article.
-
Alcohol tolerance encoding in sleep regulatory circadian neurons in Drosophila.bioRxiv [Preprint]. 2023 Feb 2:2023.01.30.526363. doi: 10.1101/2023.01.30.526363. bioRxiv. 2023. Update in: Addict Biol. 2023 Aug;28(8):e13304. doi: 10.1111/adb.13304. PMID: 36778487 Free PMC article. Updated. Preprint.
References
-
- Aso Y, Grubel K, Busch S, Friedrich AB, Siwanowicz I, Tanimoto H. The mushroom body of adult Drosophila characterized by GAL4 drivers. J Neurogenet. 2009;23(1–2):156–172. - PubMed
-
- Atkinson NS, Brenner R, Bohm RA, Yu JY, Wilbur JL. Behavioral and electrophysiological analysis of Ca-activated K-channel transgenes in Drosophila. Ann N Y Acad Sci. 1998;860:296–305. - PubMed
-
- Ausubel FM. Current Protocols in Molecular Biology. John Wiley & Sons, Inc; USA: 1994.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

