Lack of RsmA-mediated control results in constant hypervirulence, cell elongation, and hyperflagellation in Pectobacterium wasabiae
- PMID: 23372695
- PMCID: PMC3553148
- DOI: 10.1371/journal.pone.0054248
Lack of RsmA-mediated control results in constant hypervirulence, cell elongation, and hyperflagellation in Pectobacterium wasabiae
Abstract
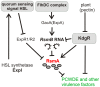
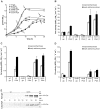
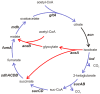
The posttranscriptional regulator RsmA controls the production of plant cell wall degrading enzymes (PCWDE) and cell motility in the Pectobacterium genus of plant pathogens. In this study the physiological role of gene regulation by RsmA is under investigation. Disruption of rsmA gene of the Pectobacterium wasabiae strain, SCC3193 resulted in 3-fold decrease in growth rate and increased virulence. The comparison of mRNA levels of the rsmA(-) mutant and wild-type using a genome-wide microarray showed, that genes responsible for successful infection, i.e. virulence factors, motility, butanediol fermentation, various secretion systems etc. were up-regulated in the rsmA(-) strain. The rsmA(-) strain exhibited a higher propensity to swarm and produce PCWDE compared to the wild-type strain. Virulence experiments in potato tubers demonstrated that in spite of its more efficient tissue maceration, the rsmA(-) strain's ability to survive within the host is reduced and the infection site is taken over by resident bacteria. Taken together, in the absence of RsmA, cells revert to a constitutively infective phenotype characterized by expression of virulence factors and swarming. We hypothesize that lack of control over these costly energetic processes results in decreased growth rate and fitness. In addition, our findings suggest a relationship between swarming and virulence in plant pathogens.
Conflict of interest statement
Figures



Similar articles
-
The global response regulator ExpA controls virulence gene expression through RsmA-mediated and RsmA-independent pathways in Pectobacterium wasabiae SCC3193.Appl Environ Microbiol. 2014 Mar;80(6):1972-84. doi: 10.1128/AEM.03829-13. Epub 2014 Jan 17. Appl Environ Microbiol. 2014. PMID: 24441162 Free PMC article.
-
Revised phylogeny and novel horizontally acquired virulence determinants of the model soft rot phytopathogen Pectobacterium wasabiae SCC3193.PLoS Pathog. 2012;8(11):e1003013. doi: 10.1371/journal.ppat.1003013. Epub 2012 Nov 1. PLoS Pathog. 2012. PMID: 23133391 Free PMC article.
-
Pectobacterium parmentieri SCC 3193 Mutants with Altered Synthesis of Cell Surface Polysaccharides Are Resistant to N4-Like Lytic Bacteriophage ϕA38 (vB_Ppp_A38) but Express Decreased Virulence in Potato (Solanum tuberosum L.) Plants.Int J Mol Sci. 2021 Jul 8;22(14):7346. doi: 10.3390/ijms22147346. Int J Mol Sci. 2021. PMID: 34298965 Free PMC article.
-
Global virulence regulation networks in phytopathogenic bacteria.Trends Microbiol. 2007 Aug;15(8):363-71. doi: 10.1016/j.tim.2007.06.005. Epub 2007 Jul 12. Trends Microbiol. 2007. PMID: 17627825 Review.
-
Regulation of host-pathogen interactions via the post-transcriptional Csr/Rsm system.Curr Opin Microbiol. 2018 Feb;41:58-67. doi: 10.1016/j.mib.2017.11.022. Epub 2017 Dec 5. Curr Opin Microbiol. 2018. PMID: 29207313 Review.
Cited by
-
Genome Wide Analysis of the Potato Soft Rot Pathogen Pectobacterium carotovorum Strain ICMP 5702 to Predict Novel Insights into Its Genetic Features.Plant Pathol J. 2022 Apr;38(2):102-114. doi: 10.5423/PPJ.OA.12.2021.0190. Epub 2022 Apr 1. Plant Pathol J. 2022. PMID: 35385916 Free PMC article.
-
Role and regulation of the Flp/Tad pilus in the virulence of Pectobacterium atrosepticum SCRI1043 and Pectobacterium wasabiae SCC3193.PLoS One. 2013 Sep 9;8(9):e73718. doi: 10.1371/journal.pone.0073718. eCollection 2013. PLoS One. 2013. PMID: 24040039 Free PMC article.
-
Multimodal Inhibition of Pectobacterium brasiliense Virulence by the Citrus Flavanone Naringenin.J Agric Food Chem. 2025 Aug 20;73(33):20792-20809. doi: 10.1021/acs.jafc.5c04312. Epub 2025 Aug 6. J Agric Food Chem. 2025. PMID: 40767039 Free PMC article.
-
Plant phenolic acids affect the virulence of Pectobacterium aroidearum and P. carotovorum ssp. brasiliense via quorum sensing regulation.Mol Plant Pathol. 2016 May;17(4):487-500. doi: 10.1111/mpp.12295. Epub 2015 Sep 7. Mol Plant Pathol. 2016. PMID: 26177258 Free PMC article.
-
The RNA-binding protein CsrA plays a central role in positively regulating virulence factors in Erwinia amylovora.Sci Rep. 2016 Nov 15;6:37195. doi: 10.1038/srep37195. Sci Rep. 2016. PMID: 27845410 Free PMC article.
References
-
- Pérombelon MCM (2002) Potato diseases caused by soft rot erwinias: an overview of pathogenesis. Plant Pathology 51: 1–12.
-
- Liu H, Coulthurst SJ, Pritchard L, Hedley PE, Ravensdale M, et al. (2008) Quorum sensing coordinates brute force and stealth modes of infection in the plant pathogen Pectobacterium atrosepticum . PLoS pathogens 4 (6) e1000093 Available: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2413422&tool=p.... Accessed 2012 Mar 9 - PMC - PubMed
-
- Andresen L, Sala E, Kõiv V, Mäe A (2010) A role for the Rcs phosphorelay in regulating expression of plant cell wall degrading enzymes in Pectobacterium carotovorum subsp. carotovorum . Microbiology 156: 1323–1334. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

