Cis-trans isomerizations of proline residues are key to bradykinin conformations
- PMID: 23373819
- PMCID: PMC3624761
- DOI: 10.1021/ja3114505
Cis-trans isomerizations of proline residues are key to bradykinin conformations
Abstract
A recent ion mobility-mass spectrometry (IM-MS) study of the nonapeptide bradykinin (BK, amino acid sequence Arg(1)-Pro(2)-Pro(3)-Gly(4)-Phe(5)-Ser(6)-Pro(7)-Phe(8)-Arg(9)) found evidence for 10 populations of conformations that depend upon the solution composition [J. Am. Chem. Soc. 2011, 133, 13810]. Here, the role of the three proline residues (Pro(2), Pro(3), and Pro(7)) in establishing these conformations is investigated using a series of seven analogue peptides in which combinations of alanine residues are substituted for prolines. IM-MS distributions of the analogue peptides, when compared to the distribution for BK, indicate the multiple structures are associated with different combinations of cis and trans forms of the three proline residues. These data are used to assign the structures to different peptide populations that are observed under various solution conditions. The assignments also show the connectivity between structures when collisional activation is used to convert one state into another.
Figures


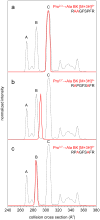
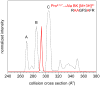
References
-
- Hall JM, Morton IKM. The Pharmacology and Immunopharmacology of Kinin Receptors. In: Farmer SG, editor. The Handbook of Immunopharmacology: The Kinin System. San Diego, CA: Academic Press; 1997. pp. 1–43.
-
- Bathon JM, Proud D. Annu Rev Pharmacol Toxicol. 1991;31:129–62. - PubMed
-
- Rocha M, Silva E, Beraldo WT, Rosenfeld G. Am J Physiol. 1949;156:261–273. - PubMed
-
- Lee SC, Rusell AF, Laidig WD. Int J Peptide Protein Res. 1990;35:367–377. - PubMed
-
- Young JK, Hicks RP. Biopolymers. 1994;34:611–623. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

