PKCα is required for inflammation-induced trafficking of extrasynaptic AMPA receptors in tonically firing lamina II dorsal horn neurons during the maintenance of persistent inflammatory pain
- PMID: 23374940
- PMCID: PMC3564511
- DOI: 10.1016/j.jpain.2012.10.015
PKCα is required for inflammation-induced trafficking of extrasynaptic AMPA receptors in tonically firing lamina II dorsal horn neurons during the maintenance of persistent inflammatory pain
Abstract
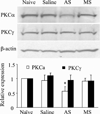
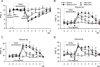
Persistent inflammation promotes internalization of synaptic GluR2-containing, Ca(2+)-impermeable AMPA receptors (AMPARs) and insertion of GluR1-containing, Ca(2+)-permeable AMPARs at extrasynaptic sites in dorsal horn neurons. Previously we have shown that internalization of synaptic GluR2-containing AMPARs requires activation of spinal cord protein kinase C alpha (PKCα), but molecular mechanisms that underlie altered trafficking of extrasynaptic AMPARs are unclear. Here, using antisense (AS) oligodeoxynucleotides (ODN) that specifically knock down PKCα, we found that a decrease in dorsal horn PKCα expression prevents complete Freund's adjuvant (CFA)-induced increase in functional expression of extrasynaptic Ca(2+)-permeable AMPARs in substantia gelatinosa (SG) neurons of the rat spinal cord. Augmented AMPA-induced currents and associated [Ca(2+)](i) transients were abolished, and the current rectification 1 day post-CFA was reversed. These changes were observed specifically in SG neurons characterized by intrinsic tonic firing properties, but not in those that exhibited strong adaptation. Finally, dorsal horn PKCα knockdown produced an antinociceptive effect on CFA-induced thermal and mechanical hypersensitivity during the maintenance period of inflammatory pain, indicating a role for PKCα in persistent inflammatory pain maintenance. Our results indicate that inflammation-induced trafficking of extrasynaptic Ca(2+)-permeable AMPARs in tonically firing SG neurons depends on PKCα, and that this PKCα-dependent trafficking may contribute to persistent inflammatory pain maintenance.
Perspective: This study shows that PKCα knockdown blocks inflammation-induced upregulation of extrasynaptic Ca(2+)-permeable AMPARs in dorsal horn neurons and produces an antinociceptive effect during the maintenance period of inflammatory pain. These findings have potential implications for use of PKCα gene-silencing therapy to prevent and/or treat persistent inflammatory pain.
Copyright © 2013 American Pain Society. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no conflict of interests.
Figures


Similar articles
-
Inflammation alters trafficking of extrasynaptic AMPA receptors in tonically firing lamina II neurons of the rat spinal dorsal horn.Pain. 2011 Apr;152(4):912-923. doi: 10.1016/j.pain.2011.01.016. Epub 2011 Feb 1. Pain. 2011. PMID: 21282008 Free PMC article.
-
Development of inflammation-induced hyperalgesia and allodynia is associated with the upregulation of extrasynaptic AMPA receptors in tonically firing lamina II dorsal horn neurons.Front Physiol. 2012 Oct 2;3:391. doi: 10.3389/fphys.2012.00391. eCollection 2012. Front Physiol. 2012. PMID: 23060815 Free PMC article.
-
Spinal cord protein interacting with C kinase 1 is required for the maintenance of complete Freund's adjuvant-induced inflammatory pain but not for incision-induced post-operative pain.Pain. 2010 Oct;151(1):226-234. doi: 10.1016/j.pain.2010.07.017. Epub 2010 Aug 8. Pain. 2010. PMID: 20696523 Free PMC article.
-
Extrasynaptic AMPA receptors in the dorsal horn: evidence and functional significance.Brain Res Bull. 2013 Apr;93:47-56. doi: 10.1016/j.brainresbull.2012.11.004. Epub 2012 Nov 26. Brain Res Bull. 2013. PMID: 23194665 Review.
-
The AMPAR subunit GluR2: still front and center-stage.Brain Res. 2000 Dec 15;886(1-2):190-207. doi: 10.1016/s0006-8993(00)02951-6. Brain Res. 2000. PMID: 11119696 Review.
Cited by
-
Inhibition of Spinal Ca(2+)-Permeable AMPA Receptors with Dicationic Compounds Alleviates Persistent Inflammatory Pain without Adverse Effects.Front Cell Neurosci. 2016 Feb 29;10:50. doi: 10.3389/fncel.2016.00050. eCollection 2016. Front Cell Neurosci. 2016. PMID: 26973464 Free PMC article.
-
Hippocalcin Promotes Neuronal Differentiation and Inhibits Astrocytic Differentiation in Neural Stem Cells.Stem Cell Reports. 2017 Jan 10;8(1):95-111. doi: 10.1016/j.stemcr.2016.11.009. Epub 2016 Dec 22. Stem Cell Reports. 2017. PMID: 28017654 Free PMC article.
-
Inflammation-induced GluA1 trafficking and membrane insertion of Ca2+ permeable AMPA receptors in dorsal horn neurons is dependent on spinal tumor necrosis factor, PI3 kinase and protein kinase A.Exp Neurol. 2017 Jul;293:144-158. doi: 10.1016/j.expneurol.2017.04.004. Epub 2017 Apr 12. Exp Neurol. 2017. PMID: 28412220 Free PMC article.
-
Molecular Mechanisms of AMPA Receptor Trafficking in the Nervous System.Int J Mol Sci. 2023 Dec 21;25(1):111. doi: 10.3390/ijms25010111. Int J Mol Sci. 2023. PMID: 38203282 Free PMC article. Review.
-
Ca2+-Permeable AMPA Receptors Contribute to Changed Dorsal Horn Neuronal Firing and Inflammatory Pain.Int J Mol Sci. 2023 Jan 25;24(3):2341. doi: 10.3390/ijms24032341. Int J Mol Sci. 2023. PMID: 36768663 Free PMC article.
References
-
- Allan SM, Rothwell NJ. Cytokines and acute neurodegeneration. Nat Rev Neurosci. 2001;2:734–744. - PubMed
-
- Atianjoh FE, Yaster M, Zhao X, Takamiya K, Xia J, Gauda EB, Huganir RL, Tao YX. Spinal cord protein interacting with C kinase 1 is required for the maintenance of complete Freund's adjuvant-induced inflammatory pain but not for incision-induced post-operative pain. Pain. 2010;151:226–234. - PMC - PubMed
-
- Barria A, Derkach V, Soderling T. Identification of the Ca2+/calmodulin-dependent protein kinase II regulatory phosphorylation site in the alpha-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate-type glutamate receptor. J Biol Chem. 1997;272:32727–32730. - PubMed
-
- Barria A, Muller D, Derkach V, Griffith LC, Soderling TR. Regulatory phosphorylation of AMPA-type glutamate receptors by CaM-KII during long-term potentiation. Science. 1997;276:2042–2045. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

