Role of impaired endothelial cell Ca(2+) signaling in uteroplacental vascular dysfunction during diabetic rat pregnancy
- PMID: 23376827
- PMCID: PMC3625897
- DOI: 10.1152/ajpheart.00513.2012
Role of impaired endothelial cell Ca(2+) signaling in uteroplacental vascular dysfunction during diabetic rat pregnancy
Abstract
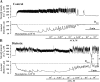
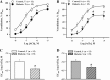
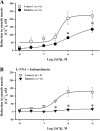
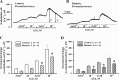
Diabetes mellitus in pregnancy is associated with impaired endothelium-mediated dilatation of maternal arteries, although the underlying cellular mechanisms remain unknown. In this study, we hypothesized that diabetes during rat gestation attenuates agonist-induced uterine vasodilation through reduced endothelial cell (EC) Ca(2+) elevations and impaired smooth muscle cell (SMC) hyperpolarization and SMC intracellular Ca(2+) concentration ([Ca(2+)]i) responses. Diabetes was induced by an injection of streptozotocin to second-day pregnant rats and confirmed by the development of maternal hyperglycemia. Control rats were injected with a citrate buffer. Fura-2-based measurements of SMC [Ca(2+)]i or microelectrode recordings of SMC membrane potential were performed concurrently with dilator responses to ACh in uteroplacental arteries from control and diabetic pregnant rats. Basal levels of EC [Ca(2+)]i and ACh-induced EC [Ca(2+)]i elevations in pressurized vessels and small EC sheets were studied as well. Diabetes reduced ACh-induced vasodilation due to a markedly impaired EDHF-mediated response. Diminished vasodilation to ACh was associated with attenuated SMC hyperpolarization and [Ca(2+)]i responses. Basal levels of EC [Ca(2+)]i and ACh-induced EC [Ca(2+)]i elevations were significantly reduced by diabetes. In conclusion, these data demonstrate that reduced endothelium-mediated hyperpolarization contributes to attenuated uteroplacental vasodilation and SMC [Ca(2+)]i responses to ACh in diabetic pregnancy. Impaired endothelial Ca(2+) signaling is in part responsible for endothelial dysfunction in the uterine resistance vasculature of diabetic rats. Pharmacological improvement of EC Ca(2+) handling may provide an important strategy for the restoration of endothelial function and enhancement of maternal blood flow in human pregnancies complicated by diabetes.
Figures



Similar articles
-
Endothelium-Dependent Hyperpolarization (EDH) in Diabetes: Mechanistic Insights and Therapeutic Implications.Int J Mol Sci. 2019 Jul 31;20(15):3737. doi: 10.3390/ijms20153737. Int J Mol Sci. 2019. PMID: 31370156 Free PMC article. Review.
-
Impairment of IKCa channels contributes to uteroplacental endothelial dysfunction in rat diabetic pregnancy.Am J Physiol Heart Circ Physiol. 2015 Aug 15;309(4):H592-604. doi: 10.1152/ajpheart.00901.2014. Epub 2015 Jun 19. Am J Physiol Heart Circ Physiol. 2015. PMID: 26092991 Free PMC article.
-
Augmented EDHF signaling in rat uteroplacental vasculature during late pregnancy.Am J Physiol Heart Circ Physiol. 2010 Nov;299(5):H1642-52. doi: 10.1152/ajpheart.00227.2010. Epub 2010 Sep 3. Am J Physiol Heart Circ Physiol. 2010. PMID: 20817830 Free PMC article.
-
Estrogen replacement enhances EDHF-mediated vasodilation of mesenteric and uterine resistance arteries: role of endothelial cell Ca2+.Am J Physiol Endocrinol Metab. 2009 Mar;296(3):E503-12. doi: 10.1152/ajpendo.90517.2008. Epub 2009 Jan 6. Am J Physiol Endocrinol Metab. 2009. PMID: 19126786 Free PMC article.
-
Potassium Channels in the Uterine Vasculature: Role in Healthy and Complicated Pregnancies.Int J Mol Sci. 2022 Aug 21;23(16):9446. doi: 10.3390/ijms23169446. Int J Mol Sci. 2022. PMID: 36012712 Free PMC article. Review.
Cited by
-
Endothelium-Dependent Hyperpolarization (EDH) in Diabetes: Mechanistic Insights and Therapeutic Implications.Int J Mol Sci. 2019 Jul 31;20(15):3737. doi: 10.3390/ijms20153737. Int J Mol Sci. 2019. PMID: 31370156 Free PMC article. Review.
-
Sex-Dependent Regulation of Placental Oleic Acid and Palmitic Acid Metabolism by Maternal Glycemia and Associations with Birthweight.Int J Mol Sci. 2022 Aug 4;23(15):8685. doi: 10.3390/ijms23158685. Int J Mol Sci. 2022. PMID: 35955818 Free PMC article.
-
The Piezo1 cation channel mediates uterine artery shear stress mechanotransduction and vasodilation during rat pregnancy.Am J Physiol Heart Circ Physiol. 2018 Oct 1;315(4):H1019-H1026. doi: 10.1152/ajpheart.00103.2018. Epub 2018 Jul 13. Am J Physiol Heart Circ Physiol. 2018. PMID: 30004235 Free PMC article.
-
Augmented dilation to nitric oxide in uterine arteries from rats with type 2 diabetes: implications for vascular adaptations to pregnancy.Am J Physiol Heart Circ Physiol. 2014 Feb 15;306(4):H610-8. doi: 10.1152/ajpheart.00588.2013. Epub 2013 Dec 13. Am J Physiol Heart Circ Physiol. 2014. PMID: 24337459 Free PMC article.
-
Impairment of IKCa channels contributes to uteroplacental endothelial dysfunction in rat diabetic pregnancy.Am J Physiol Heart Circ Physiol. 2015 Aug 15;309(4):H592-604. doi: 10.1152/ajpheart.00901.2014. Epub 2015 Jun 19. Am J Physiol Heart Circ Physiol. 2015. PMID: 26092991 Free PMC article.
References
-
- Anastasiou E, Lekakis JP, Alevizaki M, Papamichael CM, Megas J, Souvatzoglou A, Stamatelopoulos SF. Impaired endothelium-dependent vasodilatation in women with previous gestational diabetes. Diabetes Care 21: 2111–2115, 1998 - PubMed
-
- Ando J, Yamamoto K. Effects of shear stress and stretch on endothelial function. Antioxid Redox Signal 15: 1389–1403, 2011 - PubMed
-
- Ang C, Lumsden MA. Diabetes and the maternal resistance vasculature. Clin Sci (Lond) 101: 719–729, 2001 - PubMed
-
- Barden A, Singh R, Walters BN, Ritchie J, Roberman B, Beilin LJ. Factors predisposing to pre-eclampsia in women with gestational diabetes. J Hypertens 22: 2371–2378, 2004 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

