Comparing duration of response and duration of clinical benefit between fulvestrant treatment groups in the CONFIRM trial: application of new methodology
- PMID: 23378064
- PMCID: PMC3586113
- DOI: 10.1007/s10549-012-2395-8
Comparing duration of response and duration of clinical benefit between fulvestrant treatment groups in the CONFIRM trial: application of new methodology
Abstract
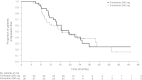
Comparisons of duration of response (DoR) and duration of clinical benefit (DoCB) within clinical trials are prone to biases. To address these biases, we used new methodology to prospectively analyze expected DoR and expected DoCB. Objective response rate and clinical benefit rate were calculated for fulvestrant 500 and 250 mg, and used to calculate expected DoR and expected DoCB for each dose group. The ratios for expected DoR and expected DoCB (expected DoR500/expected DoR250 and expected DoCB500/expected DoCB250) were then calculated, thereby allowing statistical comparisons of these endpoints between each arm of the COmparisoN of Faslodex In Recurrent or Metastatic breast cancer (CONFIRM) trial. Expected DoRs for fulvestrant 500 and 250 mg were 3.2 and 3.6 months, respectively. The expected DoR ratio between fulvestrant 500 and 250 mg was not statistically significant (0.89; 95 % CI, 0.48-1.67, P = 0.724). The expected DoCBs for fulvestrant 500 and 250 mg were 9.8 and 7.2 months, respectively. The expected DoCB ratio showed that the expected DoCB for fulvestrant 500 mg was significantly improved compared with the expected DoCB for fulvestrant 250 mg (1.36; 95 % CI, 1.07-1.73, P = 0.013). Analysis of the expected DoR and expected DoCB showed fulvestrant 500 mg significantly increased expected DoCB compared with fulvestrant 250 mg in the CONFIRM trial.
Figures

References
-
- Robertson JF, Nicholson RI, Bundred NJ, Anderson E, Rayter Z, Dowsett M, Fox JN, Gee JM, Webster A, Wakeling AE, Morris C, Dixon M. Comparison of the short-term biological effects of 7alpha-[9-(4,4,5,5,5-pentafluoropentylsulfinyl)-nonyl]estra-1,3,5, (10)-triene-3,17beta-diol (Faslodex) versus tamoxifen in postmenopausal women with primary breast cancer. Cancer Res. 2001;61:6739–6746. - PubMed
-
- DeFriend DJ, Howell A, Nicholson RI, Anderson E, Dowsett M, Mansel RE, Blamey RW, Bundred NJ, Robertson JF, Saunders C, Baum H, Walton P, Sutcliffe FA, Wakeling AE. Investigation of a new pure antiestrogen (ICI 182780) in women with primary breast cancer. Cancer Res. 1994;54:408–414. - PubMed
-
- Kuter I, Hegg R, Singer CF, Badwe R, Lowe E, on behalf of the NEWEST investigators Fulvestrant 500 versus 250 mg: first results from NEWEST, a randomized, phase II neoadjuvant trial in postmenopausal women with locally advanced, estrogen receptor-positive breast cancer. Breast Cancer Res Treat. 2008;109:589.
-
- Di Leo A, Jerusalem G, Petruzelka L, Torres R, Bondarenko IN, Khasanov R, Verhoeven D, Pedrini JL, Smirnova I, Lichinitser MR, Pendergrass K, Garnett S, Lindemann JP, Sapunar F, Martin M. Results of the CONFIRM phase III trial comparing fulvestrant 250 mg with fulvestrant 500 mg in postmenopausal women with estrogen receptor-positive advanced breast cancer. J Clin Oncol. 2010;28:4594–4600. doi: 10.1200/JCO.2010.28.8415. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

