Incorporation of aggrecan in interpenetrating network hydrogels to improve cellular performance for cartilage tissue engineering
- PMID: 23379843
- PMCID: PMC3638541
- DOI: 10.1089/ten.TEA.2012.0160
Incorporation of aggrecan in interpenetrating network hydrogels to improve cellular performance for cartilage tissue engineering
Abstract
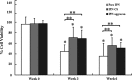
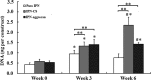
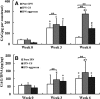
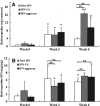
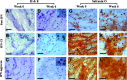
Interpenetrating network (IPN) hydrogels were recently introduced to the cartilage tissue engineering literature, with the approach of encapsulating cells in thermally gelling agarose that is then soaked in a poly(ethylene glycol) diacrylate (PEGDA) solution, which is then photopolymerized. These IPNs possess significantly enhanced mechanical performance desirable for cartilage regeneration, potentially allowing patients to return to weight-bearing activities quickly after surgical implantation. In an effort to improve cell viability and performance, inspiration was drawn from previous studies that have elicited positive chondrogenic responses to aggrecan, the proteoglycan largely responsible for the compressive stiffness of cartilage. Aggrecan was incorporated into the IPNs in conservative concentrations (40 μg/mL), and its effect was contrasted with the incorporation of chondroitin sulfate (CS), the primary glycosaminoglycan associated with aggrecan. Aggrecan was incorporated by physical entrapment within agarose and methacrylated CS was incorporated by copolymerization with PEGDA. The IPNs incorporating aggrecan or CS exhibited over 50% viability with encapsulated chondrocytes after 6 weeks. Both aggrecan and CS improved cell viability by 15.6% and 20%, respectively, relative to pure IPNs at 6 weeks culture time. In summary, we have introduced the novel approach of including a raw material from cartilage, namely aggrecan, to serve as a bioactive signal to cells encapsulated in IPN hydrogels for cartilage tissue engineering, which led to improved performance of encapsulated chondrocytes.
Figures


Similar articles
-
The bioactivity of agarose-PEGDA interpenetrating network hydrogels with covalently immobilized RGD peptides and physically entrapped aggrecan.Biomaterials. 2014 Apr;35(11):3558-70. doi: 10.1016/j.biomaterials.2014.01.002. Epub 2014 Jan 24. Biomaterials. 2014. PMID: 24462353 Free PMC article.
-
Using chondroitin sulfate to improve the viability and biosynthesis of chondrocytes encapsulated in interpenetrating network (IPN) hydrogels of agarose and poly(ethylene glycol) diacrylate.J Mater Sci Mater Med. 2012 Jan;23(1):157-70. doi: 10.1007/s10856-011-4499-9. Epub 2011 Nov 25. J Mater Sci Mater Med. 2012. PMID: 22116661 Free PMC article.
-
Hierarchically designed agarose and poly(ethylene glycol) interpenetrating network hydrogels for cartilage tissue engineering.Tissue Eng Part C Methods. 2010 Dec;16(6):1533-42. doi: 10.1089/ten.tec.2009.0761. Epub 2010 Jul 13. Tissue Eng Part C Methods. 2010. PMID: 20626274 Free PMC article.
-
Aggrecan, the Primary Weight-Bearing Cartilage Proteoglycan, Has Context-Dependent, Cell-Directive Properties in Embryonic Development and Neurogenesis: Aggrecan Glycan Side Chain Modifications Convey Interactive Biodiversity.Biomolecules. 2020 Aug 27;10(9):1244. doi: 10.3390/biom10091244. Biomolecules. 2020. PMID: 32867198 Free PMC article. Review.
-
Sulfated Hydrogels in Intervertebral Disc and Cartilage Research.Cells. 2021 Dec 17;10(12):3568. doi: 10.3390/cells10123568. Cells. 2021. PMID: 34944076 Free PMC article. Review.
Cited by
-
Bioactive Microsphere-Based Scaffolds Containing Decellularized Cartilage.Macromol Biosci. 2015 Jul;15(7):979-89. doi: 10.1002/mabi.201400472. Epub 2015 Mar 27. Macromol Biosci. 2015. PMID: 25821206 Free PMC article.
-
Recent Developments in Tough Hydrogels for Biomedical Applications.Gels. 2018 May 22;4(2):46. doi: 10.3390/gels4020046. Gels. 2018. PMID: 30674822 Free PMC article. Review.
-
An Injectable Enzymatically Crosslinked Carboxymethylated Pullulan/Chondroitin Sulfate Hydrogel for Cartilage Tissue Engineering.Sci Rep. 2016 Jan 28;6:20014. doi: 10.1038/srep20014. Sci Rep. 2016. PMID: 26817622 Free PMC article.
-
Bioactive decellularized extracellular matrix-based hydrogel supports human adipose tissue-derived stem cell maintenance and fibrocartilage phenotype.Front Bioeng Biotechnol. 2024 Jan 8;11:1304030. doi: 10.3389/fbioe.2023.1304030. eCollection 2023. Front Bioeng Biotechnol. 2024. PMID: 38260748 Free PMC article.
-
Recent progress in the manipulation of biochemical and biophysical cues for engineering functional tissues.Bioeng Transl Med. 2022 Aug 5;8(2):e10383. doi: 10.1002/btm2.10383. eCollection 2023 Mar. Bioeng Transl Med. 2022. PMID: 36925674 Free PMC article. Review.
References
-
- Elisseeff J. Puleo C. Yang F. Sharma B. Advances in skeletal tissue engineering with hydrogels. Orthod Craniofac Res. 2005;8:150. - PubMed
-
- Ifkovits J.L. Burdick J.A. Review: photopolymerizable and degradable biomaterials for tissue engineering applications. Tissue Eng. 2007;13:2369. - PubMed
-
- Vinatier C. Guicheux J. Daculsi G. Layrolle P. Weiss P. Cartilage and bone tissue engineering using hydrogels. Biomed Mater Eng. 2006;16:S107. - PubMed
-
- Awad H.A. Wickham M.Q. Leddy H.A. Gimble J.M. Guilak F. Chondrogenic differentiation of adipose-derived adult stem cells in agarose, alginate, and gelatin scaffolds. Biomaterials. 2004;25:3211. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

