Contrasting roles of dietary selenium and selenoproteins in chemically induced hepatocarcinogenesis
- PMID: 23389288
- PMCID: PMC3643414
- DOI: 10.1093/carcin/bgt011
Contrasting roles of dietary selenium and selenoproteins in chemically induced hepatocarcinogenesis
Abstract
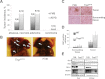
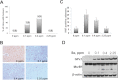
Selenium (Se) has long been known for its cancer prevention properties, but the molecular basis remains unclear. The principal questions in assessing the effect of dietary Se in cancer are whether selenoproteins, small molecule selenocompounds, or both, are involved, and under which conditions and genotypes Se may be protective. In this study, we examined diethylnitrosamine-induced hepatocarcinogenesis in mice lacking a subset of selenoproteins due to expression of a mutant selenocysteine tRNA gene (Trsp (A37G) mice). To uncouple the effects of selenocompounds and selenoproteins, these animals were examined at several levels of dietary Se. Our analysis revealed that tumorigenesis in Trsp (A37G) mice maintained on the adequate Se diet was increased. However, in the control, wild-type mice, both Se deficiency and high Se levels protected against tumorigenesis. We further found that the Se-deficient diet induced severe neurological phenotypes in Trsp A37G mice. Surprisingly, a similar phenotype could be induced in these mice at high dietary Se intake. Overall, our results show a complex role of Se in chemically induced hepatocarcinogenesis, which involves interaction among selenoproteins, selenocompounds and toxins, and depends on genotype and background of the animals.
Figures




References
-
- Brigelius-Flohé R. (2008). Selenium compounds and selenoproteins in cancer. Chem. Biodivers., 5, 389–395 - PubMed
-
- Duffield-Lillico A.J., et al. (2003). Selenium supplementation, baseline plasma selenium status and incidence of prostate cancer: an analysis of the complete treatment period of the Nutritional Prevention of Cancer Trial. BJU Int., 91, 608–612 - PubMed
-
- Duffield-Lillico A.J., et al. (2002). Baseline characteristics and the effect of selenium supplementation on cancer incidence in a randomized clinical trial: a summary report of the Nutritional Prevention of Cancer Trial. Cancer Epidemiol. Biomarkers Prev., 11, 630–639 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

