Investigating lasp-2 in cell adhesion: new binding partners and roles in motility
- PMID: 23389630
- PMCID: PMC3608507
- DOI: 10.1091/mbc.E12-10-0723
Investigating lasp-2 in cell adhesion: new binding partners and roles in motility
Abstract
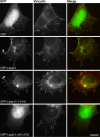
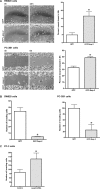
Focal adhesions are intricate protein complexes that facilitate cell attachment, migration, and cellular communication. Lasp-2 (LIM-nebulette), a member of the nebulin family of actin-binding proteins, is a newly identified component of these complexes. To gain further insights into the functional role of lasp-2, we identified two additional binding partners of lasp-2: the integral focal adhesion proteins vinculin and paxillin. Of interest, the interaction of lasp-2 with its binding partners vinculin and paxillin is significantly reduced in the presence of lasp-1, another nebulin family member. The presence of lasp-2 appears to enhance the interaction of vinculin and paxillin with each other; however, as with the interaction of lasp-2 with vinculin or paxillin, this effect is greatly diminished in the presence of excess lasp-1. This suggests that the interplay between lasp-2 and lasp-1 could be an adhesion regulatory mechanism. Lasp-2's potential role in metastasis is revealed, as overexpression of lasp-2 in either SW620 or PC-3B1 cells-metastatic cancer cell lines-increases cell migration but impedes cell invasion, suggesting that the enhanced interaction of vinculin and paxillin may functionally destabilize focal adhesion composition. Taken together, these data suggest that lasp-2 has an important role in coordinating and regulating the composition and dynamics of focal adhesions.
Figures


Similar articles
-
Paxillin and Hic-5 interaction with vinculin is differentially regulated by Rac1 and RhoA.PLoS One. 2012;7(5):e37990. doi: 10.1371/journal.pone.0037990. Epub 2012 May 22. PLoS One. 2012. PMID: 22629471 Free PMC article.
-
Zyxin interacts with the SH3 domains of the cytoskeletal proteins LIM-nebulette and Lasp-1.J Biol Chem. 2004 May 7;279(19):20401-10. doi: 10.1074/jbc.M310304200. Epub 2004 Mar 5. J Biol Chem. 2004. PMID: 15004028
-
Lasp-1 regulates podosome function.PLoS One. 2012;7(4):e35340. doi: 10.1371/journal.pone.0035340. Epub 2012 Apr 13. PLoS One. 2012. PMID: 22514729 Free PMC article.
-
Roles of Nebulin Family Members in the Heart.Circ J. 2015;79(10):2081-7. doi: 10.1253/circj.CJ-15-0854. Epub 2015 Aug 31. Circ J. 2015. PMID: 26321576 Review.
-
Paxillin and Kindlin: Research Progress and Biological Functions.Biomolecules. 2025 Jan 24;15(2):173. doi: 10.3390/biom15020173. Biomolecules. 2025. PMID: 40001476 Free PMC article. Review.
Cited by
-
LASP2 suppresses colorectal cancer progression through JNK/p38 MAPK pathway meditated epithelial-mesenchymal transition.Cell Commun Signal. 2017 Jun 12;15(1):21. doi: 10.1186/s12964-017-0179-9. Cell Commun Signal. 2017. PMID: 28606091 Free PMC article.
-
LIM and SH3 protein 1 localizes to the leading edge of protruding lamellipodia and regulates axon development.Mol Biol Cell. 2020 Nov 15;31(24):2718-2732. doi: 10.1091/mbc.E20-06-0366. Epub 2020 Sep 30. Mol Biol Cell. 2020. PMID: 32997597 Free PMC article.
-
Nebulette Expression Is Associated with Lymph Node Metastasis in Patients with Colorectal Cancer.Middle East J Dig Dis. 2018 Jul;10(3):174-179. doi: 10.15171/mejdd.2018.107. Epub 2018 May 6. Middle East J Dig Dis. 2018. PMID: 30186581 Free PMC article.
-
The Expressions and Mechanisms of Sarcomeric Proteins in Cancers.Dis Markers. 2020 Jun 30;2020:8885286. doi: 10.1155/2020/8885286. eCollection 2020. Dis Markers. 2020. PMID: 32670437 Free PMC article. Review.
-
An update on the LIM and SH3 domain protein 1 (LASP1): a versatile structural, signaling, and biomarker protein.Oncotarget. 2015 Jan 1;6(1):26-42. doi: 10.18632/oncotarget.3083. Oncotarget. 2015. PMID: 25622104 Free PMC article. Review.
References
-
- Beckerle MC. Zyxin: zinc fingers at sites of cell adhesion. Bioessays. 1997;19:949–957. - PubMed
-
- Chew CS, Chen X, Parente JA, Jr, Tarrer S, Okamoto C, Qin HY. Lasp-1 binds to non-muscle F-actin in vitro and is localized within multiple sites of dynamic actin assembly in vivo. J Cell Sci. 2002;115:4787–4799. - PubMed
-
- Crawford AW, Beckerle MC. Purification and characterization of zyxin, an 82,000-dalton component of adherens junctions. J Biol Chem. 1991;266:5847–5853. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

