The natural product betulinic acid rapidly promotes amyloid-β fibril formation at the expense of soluble oligomers
- PMID: 23401880
- PMCID: PMC3957411
- DOI: 10.1021/cn300030a
The natural product betulinic acid rapidly promotes amyloid-β fibril formation at the expense of soluble oligomers
Abstract
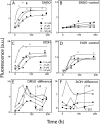
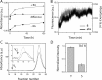
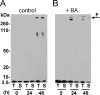
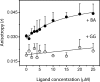
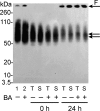
The biochemical hallmarks of Alzheimer’s disease (AD) are the aggregates of amyloid-β (Aβ) peptide that deposit in brains of AD patients as senile plaques. The monomeric Aβ undergoes aggregation in a nucleation-dependent manner to form insoluble fibrils. Emerging evidence suggests that the low-molecular-weight aggregates called “soluble oligomers” are the primary neurotoxic agents as opposed to the fibrils. Needless to say, developing Aβ aggregation inhibitors is imperative for a meaningful progress toward AD therapy. In this report, we have explored the in vitro interactions between a natural product called betulinic acid (BA) and Aβ42 peptide. BA has found its therapeutic use in several human pathologies including cancer, HIV-related AIDS, and nervous system disorders. The results from this study indicate that BA rapidly promotes the formation of Aβ42 fibrils and, in doing so, partly circumvents the formation of potentially neurotoxic soluble oligomers. Furthermore, the promotion of fibrils by BA seems to be specific for the fibril formation “on-pathway”, and it fails to interact with aggregates that are formed outside this obligatory pathway. The unique ability of BA to promote fibrils at the expense of oligomers along with its well-known pharmacological properties make BA a potential therapeutic agent for AD.
Figures

Similar articles
-
Soluble oligomers from a non-disease related protein mimic Abeta-induced tau hyperphosphorylation and neurodegeneration.J Neurochem. 2007 Oct;103(2):736-48. doi: 10.1111/j.1471-4159.2007.04809.x. Epub 2007 Aug 28. J Neurochem. 2007. PMID: 17727639
-
The polyphenol piceid destabilizes preformed amyloid fibrils and oligomers in vitro: hypothesis on possible molecular mechanisms.Neurochem Res. 2009 Jun;34(6):1120-8. doi: 10.1007/s11064-008-9883-6. Epub 2008 Nov 23. Neurochem Res. 2009. PMID: 19030989
-
New Insights into the Inhibition Mechanism of Betulinic Acid on α-Glucosidase.J Agric Food Chem. 2018 Jul 11;66(27):7065-7075. doi: 10.1021/acs.jafc.8b02992. Epub 2018 Jun 26. J Agric Food Chem. 2018. PMID: 29902001
-
[Advances in the study of structural modifications and biological activities of betulinic acids].Yao Xue Xue Bao. 2010 Nov;45(11):1339-45. Yao Xue Xue Bao. 2010. PMID: 21361032 Review. Chinese.
-
Betulin-betulinic acid natural product based analogs as anti-cancer agents.Anticancer Agents Med Chem. 2013 Dec;13(10):1477-99. doi: 10.2174/18715230113129990094. Anticancer Agents Med Chem. 2013. PMID: 23848199 Review.
Cited by
-
A Direct Relationship Between 'Blood Stasis' and Fibrinaloid Microclots in Chronic, Inflammatory, and Vascular Diseases, and Some Traditional Natural Products Approaches to Treatment.Pharmaceuticals (Basel). 2025 May 12;18(5):712. doi: 10.3390/ph18050712. Pharmaceuticals (Basel). 2025. PMID: 40430532 Free PMC article. Review.
-
The effects of betulinic acid chronic administration on the motor, non-motor behaviors, and globus pallidus local field potential power in a rat model of hemiparkinsonism.Iran J Basic Med Sci. 2022 Nov;25(11):1357-1363. doi: 10.22038/IJBMS.2022.65623.14434. Iran J Basic Med Sci. 2022. PMID: 36474564 Free PMC article.
-
Cause and consequence of Aβ - Lipid interactions in Alzheimer disease pathogenesis.Biochim Biophys Acta Biomembr. 2018 Sep;1860(9):1652-1662. doi: 10.1016/j.bbamem.2018.03.004. Epub 2018 Mar 9. Biochim Biophys Acta Biomembr. 2018. PMID: 29526709 Free PMC article. Review.
-
Ring-like N-fold Models of Aβ42 fibrils.Sci Rep. 2017 Jul 26;7(1):6588. doi: 10.1038/s41598-017-06846-0. Sci Rep. 2017. PMID: 28747632 Free PMC article.
-
Effect of Lauric Acid on the Stability of Aβ42 Oligomers.ACS Omega. 2021 Feb 17;6(8):5795-5804. doi: 10.1021/acsomega.0c06211. eCollection 2021 Mar 2. ACS Omega. 2021. PMID: 33681618 Free PMC article.
References
-
- Kawarabayashi T.; Shoji M.; Younkin L. H.; Wen-Lang L.; Dickson D. W.; Murakami T.; Matsubara E.; Abe K.; Ashe K. H.; Younkin S. G. (2004) Dimeric amyloid beta protein rapidly accumulates in lipid rafts followed by apolipoprotein E and phosphorylated tau accumulation in the Tg2576 mouse model of Alzheimer’s disease. J. Neurosci. 24, 3801–3809. - PMC - PubMed
-
- Lesne S.; Koh M. T.; Kotilinek L.; Kayed R.; Glabe C. G.; Yang A.; Gallagher M.; Ashe K. H. (2006) A specific amyloid-beta protein assembly in the brain impairs memory. Nature 440, 352–357. - PubMed
-
- Shankar G. M.; Li S.; Mehta T. H.; Garcia-Munoz A.; Shepardson N. E.; Smith I.; Brett F. M.; Farrell M. A.; Rowan M. J.; Lemere C. A.; Regan C. M.; Walsh D. M.; Sabatini B. L.; Selkoe D. J. (2008) Amyloid-beta protein dimers isolated directly from Alzheimer’s brains impair synaptic plasticity and memory. Nat. Med. 14, 837–842. - PMC - PubMed
-
- Goldsbury C.; Frey P.; Olivieri V.; Aebi U.; Muller S. A. (2005) Multiple assembly pathways underlie amyloid-beta fibril polymorphisms. J. Mol. Biol. 352, 282–298. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

