Effect of short-term hyperglycemia on protein kinase C alpha activation in human erythrocytes
- PMID: 23403705
- PMCID: PMC3700022
- DOI: 10.1900/RDS.2012.9.94
Effect of short-term hyperglycemia on protein kinase C alpha activation in human erythrocytes
Abstract
Background: Diabetes mellitus, characterized by chronic hyperglycemia, is known to have a deleterious effect on erythrocyte structure and hemodynamic characteristics, which eventually contribute to diabetes-associated vascular complications. Protein kinase C alpha (PKCα) is a major regulator of many metabolic processes and structural changes in erythrocytes, and may play a significant role in the development of hyperglycemia-mediated cellular abnormalities.
Aim: We hypothesized that acute hyperglycemic stress may affect erythrocyte structure and metabolic properties through its effect on PKCα membrane content and activity.
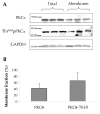
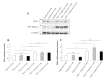
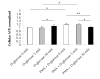
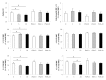
Results: Erythrocytes, from healthy individuals acutely exposed to a glucose enriched media, showed a significant decrease in the membranous fraction of PKCα and its phosphorylation (p = 0.005 and p = 0.0004, respectively). These alterations correlated with decreased affinity of PKCα to its membrane substrates (4.1R and GLUT1) and reduced RBC deformability (p = 0.017). Pre-activation of erythrocytes with PKC activator, PMA, minimized the effect of glucose on the membrane PKCα fraction and RBC deformability (p > 0.05).
Conclusions: Acute glycemia-induced inhibition of PKCα membranous translocation and activation is associated with reduced erythrocyte membrane deformability.
Figures

Similar articles
-
Mechanism of inhibition of sequestration of protein kinase C alpha/betaII by ceramide. Roles of ceramide-activated protein phosphatases and phosphorylation/dephosphorylation of protein kinase C alpha/betaII on threonine 638/641.J Biol Chem. 2007 Jul 13;282(28):20647-56. doi: 10.1074/jbc.M609162200. Epub 2007 May 15. J Biol Chem. 2007. PMID: 17504762
-
Phorbol 12-myristate 13-acetate-stimulated phosphorylation of erythrocyte membrane skeletal proteins is blocked by calpain inhibitors: possible role of protein kinase M.Biochem J. 1993 Dec 15;296 ( Pt 3)(Pt 3):675-83. doi: 10.1042/bj2960675. Biochem J. 1993. PMID: 8280066 Free PMC article.
-
Protein kinase C mediates erythrocyte "programmed cell death" following glucose depletion.Am J Physiol Cell Physiol. 2006 Jan;290(1):C244-53. doi: 10.1152/ajpcell.00283.2005. Am J Physiol Cell Physiol. 2006. PMID: 16338977
-
Erythrocyte deformability and its variation in diabetes mellitus.Indian J Exp Biol. 2007 Jan;45(1):121-8. Indian J Exp Biol. 2007. PMID: 17249336 Review.
-
The role of protein kinase C activation and the vascular complications of diabetes.Pharmacol Res. 2007 Jun;55(6):498-510. doi: 10.1016/j.phrs.2007.04.016. Epub 2007 May 5. Pharmacol Res. 2007. PMID: 17574431 Review.
Cited by
-
Correlation of Hemoglobin A1c with Red Cell Width Distribution and Other Parameters of Red Blood Cells in Type II Diabetes Mellitus.Cureus. 2019 Aug 30;11(8):e5533. doi: 10.7759/cureus.5533. Cureus. 2019. PMID: 31687307 Free PMC article.
-
The Relationship between Red Blood Cell Distribution Width and Incident Diabetes in Chinese Adults: A Cohort Study.J Diabetes Res. 2020 Feb 27;2020:1623247. doi: 10.1155/2020/1623247. eCollection 2020. J Diabetes Res. 2020. PMID: 32185232 Free PMC article.
-
ICln: a new regulator of non-erythroid 4.1R localisation and function.PLoS One. 2014 Oct 8;9(10):e108826. doi: 10.1371/journal.pone.0108826. eCollection 2014. PLoS One. 2014. PMID: 25295618 Free PMC article.
-
Diabetic foot disease is associated with reduced erythrocyte deformability.Int Wound J. 2016 Aug;13(4):500-4. doi: 10.1111/iwj.12466. Epub 2015 May 28. Int Wound J. 2016. PMID: 26018868 Free PMC article.
-
Red blood cell distribution width and the risk of being in poor glycemic control among patients with established type 2 diabetes.Ther Clin Risk Manag. 2018 Feb 14;14:265-273. doi: 10.2147/TCRM.S155753. eCollection 2018. Ther Clin Risk Manag. 2018. PMID: 29497303 Free PMC article.
References
-
- Wong WT, Wong SL, Tian XY, Huang Y. Endothelial dysfunction: the common consequence in diabetes and hypertension. J Cardiovasc Pharmacol. 2010;55:300–307. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
