Notch2 is required in somatic cells for breakdown of ovarian germ-cell nests and formation of primordial follicles
- PMID: 23406467
- PMCID: PMC3606475
- DOI: 10.1186/1741-7007-11-13
Notch2 is required in somatic cells for breakdown of ovarian germ-cell nests and formation of primordial follicles
Abstract
Background: In the mouse ovary, oocytes initially develop in clusters termed germ-cell nests. Shortly after birth, these germ-cell nests break apart, and the oocytes individually become surrounded by somatic granulosa cells to form primordial follicles. Notch signaling plays essential roles during oogenesis in Drosophila, and recent studies have suggested that Notch signaling also plays an essential role during oogenesis and ovary development in mammals. However, no in vivo loss-of-function studies have been performed to establish whether Notch family receptors have an essential physiological role during normal ovarian development in mutant mice.
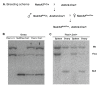
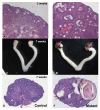
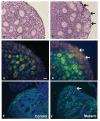
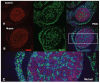
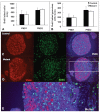
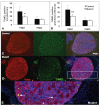
Results: Female mice with conditional deletion of the Notch2 gene in somatic granulosa cells of the ovary exhibited reduced fertility, accompanied by the formation of multi-oocyte follicles, which became hemorrhagic by 7 weeks of age. Formation of multi-oocyte follicles resulted from defects in breakdown of the primordial germ-cell nests. The ovaries of the Notch2 conditional mutant mice had increased numbers of oocytes, but decreased numbers of primordial follicles. Oocyte numbers in the Notch2 conditional mutants were increased not by excess or extended cellular proliferation, but as a result of decreased oocyte apoptosis.
Conclusions: Our work demonstrates that Notch2-mediated signaling in the somatic-cell lineage of the mouse ovary regulates oocyte apoptosis non-cell autonomously, and is essential for regulating breakdown of germ-cell nests and formation of primordial follicles. This model provides a new resource for studying the developmental and physiological roles of Notch signaling during mammalian reproductive biology.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

