BRG1 promotes COUP-TFII expression and venous specification during embryonic vascular development
- PMID: 23406903
- PMCID: PMC3585661
- DOI: 10.1242/dev.087379
BRG1 promotes COUP-TFII expression and venous specification during embryonic vascular development
Abstract
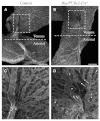
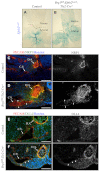
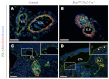
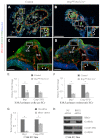
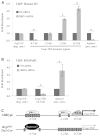
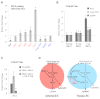
Arteries and veins acquire distinct molecular identities prior to the onset of embryonic blood circulation, and their specification is crucial for vascular development. The transcription factor COUP-TFII currently functions at the top of a signaling pathway governing venous fate. It promotes venous identity by inhibiting Notch signaling and subsequent arterialization of endothelial cells, yet nothing is known about what regulates COUP-TFII expression in veins. We now report that the chromatin-remodeling enzyme BRG1 promotes COUP-TFII expression in venous endothelial cells during murine embryonic development. Conditional deletion of Brg1 from vascular endothelial cells resulted in downregulated COUP-TFII expression and aberrant expression of arterial markers on veins. BRG1 promotes COUP-TFII expression by binding conserved regulatory elements within the COUP-TFII promoter and remodeling chromatin to make the promoter accessible to transcriptional machinery. This study provides the first description of a factor promoting COUP-TFII expression in vascular endothelium and highlights a novel role for chromatin remodeling in venous specification.
Figures

Similar articles
-
COUP-TFII is a major regulator of cell cycle and Notch signaling pathways.Mol Endocrinol. 2012 Aug;26(8):1268-77. doi: 10.1210/me.2011-1305. Epub 2012 Jun 25. Mol Endocrinol. 2012. PMID: 22734039 Free PMC article.
-
Venous Endothelial Marker COUP-TFII Regulates the Distinct Pathologic Potentials of Adult Arteries and Veins.Sci Rep. 2015 Nov 5;5:16193. doi: 10.1038/srep16193. Sci Rep. 2015. PMID: 26537113 Free PMC article.
-
Suppression of Notch signalling by the COUP-TFII transcription factor regulates vein identity.Nature. 2005 May 5;435(7038):98-104. doi: 10.1038/nature03511. Nature. 2005. PMID: 15875024
-
Arterial-venous specification during development.Circ Res. 2009 Mar 13;104(5):576-88. doi: 10.1161/CIRCRESAHA.108.188805. Circ Res. 2009. PMID: 19286613 Review.
-
Specification of arterial, venous, and lymphatic endothelial cells during embryonic development.Histol Histopathol. 2010 May;25(5):637-46. doi: 10.14670/HH-25.637. Histol Histopathol. 2010. PMID: 20238301 Free PMC article. Review.
Cited by
-
BRG1 attenuates colonic inflammation and tumorigenesis through autophagy-dependent oxidative stress sequestration.Nat Commun. 2019 Oct 10;10(1):4614. doi: 10.1038/s41467-019-12573-z. Nat Commun. 2019. PMID: 31601814 Free PMC article.
-
SWI/SNF chromatin-remodeling complexes in cardiovascular development and disease.Cardiovasc Pathol. 2014 Mar-Apr;23(2):85-91. doi: 10.1016/j.carpath.2013.09.003. Epub 2013 Oct 4. Cardiovasc Pathol. 2014. PMID: 24183004 Free PMC article. Review.
-
Ezh2-mediated repression of a transcriptional pathway upstream of Mmp9 maintains integrity of the developing vasculature.Development. 2014 Dec;141(23):4610-7. doi: 10.1242/dev.112607. Epub 2014 Oct 30. Development. 2014. PMID: 25359725 Free PMC article.
-
Molecular identity of arteries, veins, and lymphatics.J Vasc Surg. 2019 Jan;69(1):253-262. doi: 10.1016/j.jvs.2018.06.195. Epub 2018 Aug 25. J Vasc Surg. 2019. PMID: 30154011 Free PMC article. Review.
-
Isoforms of the orphan nuclear receptor COUP‑TFII differentially modulate pancreatic cancer progression.Int J Oncol. 2022 May;60(5):55. doi: 10.3892/ijo.2022.5345. Epub 2022 Mar 29. Int J Oncol. 2022. PMID: 35348189 Free PMC article.
References
-
- Adams R. H., Wilkinson G. A., Weiss C., Diella F., Gale N. W., Deutsch U., Risau W., Klein R. (1999). Roles of ephrinB ligands and EphB receptors in cardiovascular development: demarcation of arterial/venous domains, vascular morphogenesis, and sprouting angiogenesis. Genes Dev. 13, 295–306 - PMC - PubMed
-
- Brault V., Moore R., Kutsch S., Ishibashi M., Rowitch D. H., McMahon A. P., Sommer L., Boussadia O., Kemler R. (2001). Inactivation of the beta-catenin gene by Wnt1-Cre-mediated deletion results in dramatic brain malformation and failure of craniofacial development. Development 128, 1253–1264 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

