Bryostatin-1, Fenretinide and 1α,25 (OH)(2)D(3) Induce Growth Inhibition, Apoptosis and Differentiation in T and B Cell-Derived Acute Lymphoblastic Leukemia Cell Lines (CCRF-CEM and Nalm-6)
- PMID: 23407583
- PMCID: PMC3558194
Bryostatin-1, Fenretinide and 1α,25 (OH)(2)D(3) Induce Growth Inhibition, Apoptosis and Differentiation in T and B Cell-Derived Acute Lymphoblastic Leukemia Cell Lines (CCRF-CEM and Nalm-6)
Abstract
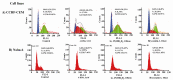
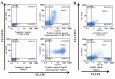
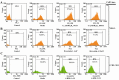
In many acute leukemias, normal differentiation does not occur. However, in many cell lines derived from hematologic malignancies, differentiation or apoptosis can be induced by variety of agents. Despite advances in the treatment of Acute Lymphoblastic Leukemia (ALL), in most patients long-term survival rates remain unsatisfactory, especially in T-cell derived ALL. Thus we studied the anti-cancer effects of fenretinide, 1α,25(OH)(2)D(3), and bryostatin-1 in CCRF-CEM (T-cell derived) and Nalm-6 (B-cell derived) ALL cell lines. Using MTT assays, both cell lines were shown to exhibit increased inhibition of proliferation at micro (fenretinide) and nanomolar (1α,25(OH)(2)D(3), bryostatin-1) concentrations. These anti-cancer agents were shown to induce apoptosis and activate caspase-3 pathway in both ALL cell lines. Furthermore, for the first time we are reporting consistent anti-proliferative and apoptotic effects of Bryostatin-1 in ALL T-cell derived cell line with the lowest ED(50) (ranging 4.6-7.4 nM). To evaluate the differentiation induction by fenretinide, 1α,25(OH)(2)D(3), and bryostatin-1 in ALL cell lines, we assayed for the expressions of CD19, CD38 markers on Nalm-6 and CD7 marker on CCRF-CEM cell line. The flow cytometric analysis showed a significant increase in expression of CD markers in response to anti-cancer drug treatments. To assay the effects of anti-cancer drugs on cell cycle distribution, cell cycle analysis using flow cytometry was employed. These anti-cancer drugs appear to affect the CCRF-CEM and Nalm-6 cell cycles differently (G0/G1 and G2/M arrest, respectively). Overall results demonstrate that the anti-cancer agents used in this study are strong inhibitors of ALL cell proliferation and inducers of apoptosis and differentiation in vitro. These findings may be quite helpful if these drugs are to be used for differentiation therapy of ALL patients in clinics in the future. Further studies are warranted to establish the in vivo effect of these drugs particularly in patients with T-cell derived ALL.
Keywords: Acute lymphoblastic leukemia; Apoptosis; Cell differentiation; Flow cytometry.
Figures



References
-
- Estey E. Treatment of relapsed and refractory acute myelogenous leukemia. Leukemia. 2000;14(3):476–479. - PubMed
-
- Cheok MH, Evans WE. Acute lymphoblastic leukaemia: a model for the pharmacogenomics of cancer therapy. Nat Rev Cancer. 2006;6(2):117–129. - PubMed
-
- Miller WH, Jr, Waxman S. Differentiation induction as a treatment for hematologic malignancies. Oncogene. 2002;21(21):3496–3506. - PubMed
-
- Greenberg ER, Sporn MB. Antioxidant vitamins, cancer, and cardiovascular disease. N Engl J Med. 1996;334(18):1189–1190. - PubMed
-
- Reynolds CP, Lemons RS. Retinoid therapy of childhood cancer. Hematol Oncol Clin North Am. 2001;15(5):867–910. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
