Expression and Single-step Purification of GRA8 Antigen of Toxoplasma gondii in Escherichia coli
- PMID: 23407862
- PMCID: PMC3558177
Expression and Single-step Purification of GRA8 Antigen of Toxoplasma gondii in Escherichia coli
Abstract
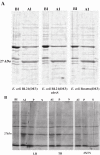
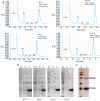
Diagnosis of Toxoplasma gondii (T.gondii) infection is of great medical importance especially for pregnant women and immunosuppressed patients. Numerous studies have shown that the recombinant production of several toxoplasma antigens, including dense granule antigens (GRAs) has a great potential as diagnostic reagents. Previous studies reported expression of amino terminal GRA8 protein in fusion with large tags. In the present study, we produced truncated GRA8 (GRA8), excluded from the signal peptide and C-terminal transmembrane domain, with a short fusion tag in Escherichia coli (E.coli). GRA8 was purified using an optimized single-step Immobilized Metal ion Affinity Chromatography (IMAC). The purity and yield of GRA8 was highest at pH = 9.25. At this pH, 13.6 mg of GRA8 was obtained with the purity of 97.97%. Immunogenicity of the protein was evaluated in Western blot analysis showing the serum sample from a rabbit immunized with GRA8 recognized a single antigen of T.gondii tachyzoite at the expected molecular weight of native GRA8. To diagnosis acute toxoplasma infection in pregnant women, an indirect immunoglobulin M (IgM) enzyme-linked immunosorbent assay (ELISA) was developed using GRA8 resulting in a test specificity and sensitivity of 97.1% and 60.6%, respectively. These results demonstrated that immunogenic GRA8 can be produced in fusion with a short tag and purified near to homogeneity using an optimized IMAC. GRA8-IgM-ELISA was useful for detection of acute toxoplasma infection.
Keywords: Enzyme-linked immunosorbent assay; GRA8 protein; Gene expression; Immunoglobulin M; Toxoplasma gondii.
Figures

Similar articles
-
Bacterial production of dense granule antigen GRA8 of Toxoplasma gondii.Iran Biomed J. 2009 Jul;13(3):145-51. Iran Biomed J. 2009. PMID: 19688020
-
Development and validation of a time-resolved fluorescence immunoassay for the detection of anti-Toxoplasma gondii antibodies in goats.Vet Parasitol. 2021 May;293:109432. doi: 10.1016/j.vetpar.2021.109432. Epub 2021 Apr 21. Vet Parasitol. 2021. PMID: 33915322
-
Development and evaluation of indirect enzyme-linked immunosorbent assay using recombinant dense granule antigen 7 protein for the detection of Toxoplasma gondii infection in cats in Thailand.Vet World. 2022 Mar;15(3):602-610. doi: 10.14202/vetworld.2022.602-610. Epub 2022 Mar 12. Vet World. 2022. PMID: 35497967 Free PMC article.
-
A novel luciferase-linked antibody capture assay (LACA) for the diagnosis of Toxoplasma gondii infection in chickens.Parasitol Int. 2020 Aug;77:102125. doi: 10.1016/j.parint.2020.102125. Epub 2020 Apr 18. Parasitol Int. 2020. PMID: 32311471
-
[Expression of truncated Toxoplasma gondii GRA8 in the prokaryotic expression plasmids].Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2005 Jun 30;23(3):129-34. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2005. PMID: 16299997 Chinese.
Cited by
-
Assessment of Recombinant A2-Latex Agglutination Test (RA2-LAT) and RA2-ELISA for Detection of Canine Visceral Leishmaniasis: A Comparative Field Study with Direct Agglutination Test in Northwestern Iran.Iran J Parasitol. 2018 Apr-Jun;13(2):172-179. Iran J Parasitol. 2018. PMID: 30069200 Free PMC article.
-
Development and evaluation of recombinant GRA8 protein for the serodiagnosis of Toxoplasma gondii infection in goats.BMC Vet Res. 2021 Jan 9;17(1):27. doi: 10.1186/s12917-020-02719-3. BMC Vet Res. 2021. PMID: 33422085 Free PMC article.
-
Exposure to Toxoplasma gondii in Asian Elephants (Elephas maximus indicus) in Thailand.Pathogens. 2021 Dec 21;11(1):2. doi: 10.3390/pathogens11010002. Pathogens. 2021. PMID: 35055950 Free PMC article.
-
IgM Antibody Detection as a Diagnostic Marker for Acute Toxoplasmosis: Current Status of Studies and Main Limitations.Antibodies (Basel). 2025 May 21;14(2):44. doi: 10.3390/antib14020044. Antibodies (Basel). 2025. PMID: 40407696 Free PMC article. Review.
-
Evaluation of Protective Immune Response Induced by a DNA Vaccine Encoding GRA8 against Acute Toxoplasmosis in a Murine Model.Korean J Parasitol. 2018 Aug;56(4):325-334. doi: 10.3347/kjp.2018.56.4.325. Epub 2018 Aug 31. Korean J Parasitol. 2018. PMID: 30196664 Free PMC article.
References
-
- Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet. 2004;363:1965–1976. - PubMed
-
- Kotresha D, Noordin R. Recombinant proteins in the diagnosis of toxoplasmosis. APMIS. 2010;118(8):529–542. - PubMed
-
- Sukthana Y. Toxoplasmosis: beyond animals to humans. Trends Parasitol. 2006;22:137–142. - PubMed
-
- Wallon M, Kodjikian L, Binquet C, Garweg J, Fleury J, Quantin C, et al. Long-term ocular prognosis in 327 children with congenital toxoplasmosis. Pediatrics. 2004;113(6):1567–1572. - PubMed
LinkOut - more resources
Full Text Sources
