Inhibition of Ca²⁺/calmodulin-dependent protein kinase kinase 2 stimulates osteoblast formation and inhibits osteoclast differentiation
- PMID: 23408651
- PMCID: PMC3688641
- DOI: 10.1002/jbmr.1890
Inhibition of Ca²⁺/calmodulin-dependent protein kinase kinase 2 stimulates osteoblast formation and inhibits osteoclast differentiation
Abstract
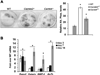
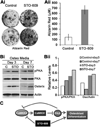
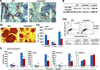
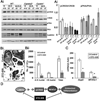
Bone remodeling, a physiological process characterized by bone formation by osteoblasts (OBs) and resorption of preexisting bone matrix by osteoclasts (OCs), is vital for the maintenance of healthy bone tissue in adult humans. Imbalances in this vital process result in pathological conditions including osteoporosis. Owing to its initial asymptomatic nature, osteoporosis is often detected only after the patient has sustained significant bone loss or a fracture. Hence, anabolic therapeutics that stimulate bone accrual is in high clinical demand. Here we identify Ca²⁺/calmodulin (CaM)-dependent protein kinase kinase 2 (CaMKK2) as a potential target for such therapeutics because its inhibition enhances OB differentiation and bone growth and suppresses OC differentiation. Mice null for CaMKK2 possess higher trabecular bone mass in their long bones, along with significantly more OBs and fewer multinuclear OCs. In vitro, although Camkk2⁻/⁻ mesenchymal stem cells (MSCs) yield significantly higher numbers of OBs, bone marrow cells from Camkk2⁻/⁻ mice produce fewer multinuclear OCs. Acute inhibition of CaMKK2 by its selective, cell-permeable pharmacological inhibitor STO-609 also results in increased OB and diminished OC formation. Further, we find phospho-protein kinase A (PKA) and Ser¹³³ phosphorylated form of cyclic adenosine monophosphate (cAMP) response element binding protein (pCREB) to be markedly elevated in OB progenitors deficient in CaMKK2. On the other hand, genetic ablation of CaMKK2 or its pharmacological inhibition in OC progenitors results in reduced pCREB as well as significantly reduced levels of its transcriptional target, nuclear factor of activated T cells, cytoplasmic (NFATc1). Moreover, in vivo administration of STO-609 results in increased OBs and diminished OCs, conferring significant protection from ovariectomy (OVX)-induced osteoporosis in adult mice. Overall, our findings reveal a novel function for CaMKK2 in bone remodeling and highlight the potential for its therapeutic inhibition as a valuable bone anabolic strategy that also inhibits OC differentiation in the treatment of osteoporosis.
Copyright © 2013 American Society for Bone and Mineral Research.
Conflict of interest statement
The authors further state that there are no restrictions on full access for all authors to all raw data, statistical analyses and material used in the study reported in this manuscript.
Figures



References
-
- Harada S, Rodan GA. Control of osteoblast function and regulation of bone mass. Nature. 2003;423(6937):349–355. - PubMed
-
- Xia S-L, Ferrier J. Localized calcium signaling in multinucleated osteoclasts. Journal of cellular physiology. 1996;167(1):148–155. - PubMed
-
- Ghosh A, Greenberg ME. Calcium signaling in neurons: molecular mechanisms and cellular consequences. Science. 1995;268(5208):239–247. - PubMed
-
- Lu KP, Means AR. Regulation of the cell cycle by calcium and calmodulin. Endocr Rev. 1993;14(1):40–58. - PubMed
-
- Colomer J, Means AR. Physiological roles of the Ca2+/CaM-dependent protein kinase cascade in health and disease. Subcell Biochem. 2007;45:169–214. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

