Human neonatal naive CD4+ T cells have enhanced activation-dependent signaling regulated by the microRNA miR-181a
- PMID: 23408835
- PMCID: PMC3952015
- DOI: 10.4049/jimmunol.1202534
Human neonatal naive CD4+ T cells have enhanced activation-dependent signaling regulated by the microRNA miR-181a
Abstract
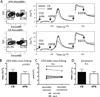
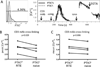
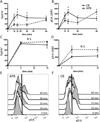
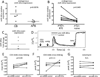
Compared with older children and adults, human neonates have reduced and delayed CD4(+) T cell immunity to certain pathogens, but the mechanisms for these developmental differences in immune function remain poorly understood. We investigated the hypothesis that impaired human neonatal CD4(+) T cell immunity was due to reduced signaling by naive CD4(+) T cells following engagement of the αβ-TCR/CD3 complex and CD28. Surprisingly, calcium flux following engagement of CD3 was significantly higher in neonatal naive CD4(+) T cells from umbilical cord blood (CB) compared with naive CD4(+) T cells from adult peripheral blood. Enhanced calcium flux was also observed in adult CD4(+) recent thymic emigrants. Neonatal naive CD4(+) T cells also had higher activation-induced Erk phosphorylation. The microRNA miR-181a, which enhances activation-induced calcium flux in murine thymocytes, was expressed at significantly higher levels in CB naive CD4(+) T cells compared with adult cells. Overexpression of miR-181a in adult naive CD4(+) T cells increased activation-induced calcium flux, implying that the increased miR-181a levels of CB naive CD4(+) T cells contributed to their enhanced signaling. In contrast, AP-1-dependent transcription, which is downstream of Erk and required for full T cell activation, was decreased in CB naive CD4(+) T cells compared with adult cells. Thus, CB naive CD4(+) T cells have enhanced activation-dependent calcium flux, indicative of the retention of a thymocyte-like phenotype. Enhanced calcium signaling and Erk phosphorylation are decoupled from downstream AP-1-dependent transcription, which is reduced and likely contributes to limitations of human fetal and neonatal CD4(+) T cell immunity.
Figures

Similar articles
-
Decreased CD154 expression by neonatal CD4+ T cells is due to limitations in both proximal and distal events of T cell activation.Int Immunol. 2003 Dec;15(12):1461-72. doi: 10.1093/intimm/dxg145. Int Immunol. 2003. PMID: 14645155
-
Impaired allogeneic activation and T-helper 1 differentiation of human cord blood naive CD4 T cells.Biol Blood Marrow Transplant. 2006 Feb;12(2):160-71. doi: 10.1016/j.bbmt.2005.10.027. Biol Blood Marrow Transplant. 2006. PMID: 16443514
-
IL-6 plays a unique role in initiating c-Maf expression during early stage of CD4 T cell activation.J Immunol. 2005 Mar 1;174(5):2720-9. doi: 10.4049/jimmunol.174.5.2720. J Immunol. 2005. PMID: 15728480
-
Posttranscriptional Gene Regulation of T Follicular Helper Cells by RNA-Binding Proteins and microRNAs.Front Immunol. 2018 Jul 31;9:1794. doi: 10.3389/fimmu.2018.01794. eCollection 2018. Front Immunol. 2018. PMID: 30108596 Free PMC article. Review.
-
Maturation of human neonatal CD4+ and CD8+ T lymphocytes into Th1/Th2 effectors.Vaccine. 1998 Aug-Sep;16(14-15):1415-9. doi: 10.1016/s0264-410x(98)00101-7. Vaccine. 1998. PMID: 9711781 Review.
Cited by
-
MicroRNA in T-Cell Development and T-Cell Mediated Acute Graft-Versus-Host Disease.Front Immunol. 2018 May 7;9:992. doi: 10.3389/fimmu.2018.00992. eCollection 2018. Front Immunol. 2018. PMID: 29867969 Free PMC article. Review.
-
MiR-142-3p attenuates the migration of CD4⁺ T cells through regulating actin cytoskeleton via RAC1 and ROCK2 in arteriosclerosis obliterans.PLoS One. 2014 Apr 17;9(4):e95514. doi: 10.1371/journal.pone.0095514. eCollection 2014. PLoS One. 2014. PMID: 24743945 Free PMC article.
-
Vaccines against respiratory viral pathogens for use in neonates: opportunities and challenges.J Immunol. 2014 Dec 1;193(11):5363-9. doi: 10.4049/jimmunol.1401410. J Immunol. 2014. PMID: 25411431 Free PMC article. Review.
-
A Genetic Model Reveals Biological Features of Neonatal CD4 Helper Cells Undergone Homeostasis in Mice.Front Cell Dev Biol. 2021 Mar 11;9:659744. doi: 10.3389/fcell.2021.659744. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33777965 Free PMC article.
-
Higher Frequency and Increased Expression of Molecules Associated with Suppression on T Regulatory Cells from Newborn Compared with Adult Nonhuman Primates.J Immunol. 2020 Oct 15;205(8):2128-2136. doi: 10.4049/jimmunol.2000461. Epub 2020 Sep 2. J Immunol. 2020. PMID: 32878911 Free PMC article.
References
-
- Lewis DB, Wilson CB. Developmental Immunology and Role of Host Defenses in Fetal and Neonatal Susceptibility to Infection. In: Klein JO, Maldonado Y, Nizet V, Remington JS, Wilson CB, editors. Infectious Diseases of the Fetus and Newborn. 7th ed. Philadelphia: Elsevier Saunders; 2011. pp. 80–191.
-
- Burchett SK, Corey L, Mohan KM, Westall J, Ashley R, Wilson CB. Diminished interferon-gamma production and lymphocyte proliferation in neonatal and postpartum primary herpes simplex virus infection. J. Infect. Dis. 1992;165:813–818. - PubMed
-
- Sullender WM, Miller JL, Yasukawa LL, Bradley JS, Black SB, Yeager AS, Arvin AM. Humoral and cell-mediated immunity in neonates with herpes simplex virus infection. J. Infect.Dis. 1987;155:28–37. - PubMed
-
- Smith S, Jacobs R, Wilson C. Immunobiology of childhood tuberculosis: A window on the ontogeny of cellular immunity. J Pediatr. 1997;131:16–26. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

