A Toxoplasma palmitoyl acyl transferase and the palmitoylated armadillo repeat protein TgARO govern apical rhoptry tethering and reveal a critical role for the rhoptries in host cell invasion but not egress
- PMID: 23408890
- PMCID: PMC3567180
- DOI: 10.1371/journal.ppat.1003162
A Toxoplasma palmitoyl acyl transferase and the palmitoylated armadillo repeat protein TgARO govern apical rhoptry tethering and reveal a critical role for the rhoptries in host cell invasion but not egress
Abstract
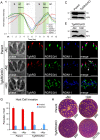
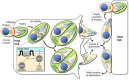
Apicomplexans are obligate intracellular parasites that actively penetrate their host cells to create an intracellular niche for replication. Commitment to invasion is thought to be mediated by the rhoptries, specialized apical secretory organelles that inject a protein complex into the host cell to form a tight-junction for parasite entry. Little is known about the molecular factors that govern rhoptry biogenesis, their subcellular organization at the apical end of the parasite and subsequent release of this organelle during invasion. We have identified a Toxoplasma palmitoyl acyltransferase, TgDHHC7, which localizes to the rhoptries. Strikingly, conditional knockdown of TgDHHC7 results in dispersed rhoptries that fail to organize at the apical end of the parasite and are instead scattered throughout the cell. While the morphology and content of these rhoptries appears normal, failure to tether at the apex results in a complete block in host cell invasion. In contrast, attachment and egress are unaffected in the knockdown, demonstrating that the rhoptries are not required for these processes. We show that rhoptry targeting of TgDHHC7 requires a short, highly conserved C-terminal region while a large, divergent N-terminal domain is dispensable for both targeting and function. Additionally, a point mutant lacking a key residue predicted to be critical for enzyme activity fails to rescue apical rhoptry tethering, strongly suggesting that tethering of the organelle is dependent upon TgDHHC7 palmitoylation activity. We tie the importance of this activity to the palmitoylated Armadillo Repeats-Only (TgARO) rhoptry protein by showing that conditional knockdown of TgARO recapitulates the dispersed rhoptry phenotype of TgDHHC7 knockdown. The unexpected finding that apicomplexans have exploited protein palmitoylation for apical organelle tethering yields new insight into the biogenesis and function of rhoptries and may provide new avenues for therapeutic intervention against Toxoplasma and related apicomplexan parasites.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
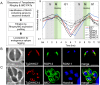


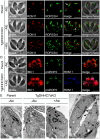
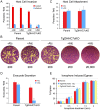
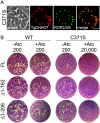
References
-
- Miller LH, Baruch DI, Marsh K, Doumbo OK (2002) The pathogenic basis of malaria. Nature 415: 673–679. - PubMed
-
- Carruthers V, Boothroyd JC (2007) Pulling together: an integrated model of Toxoplasma cell invasion. Curr Opin Microbiol 10: 83–89. - PubMed
-
- Keeley A, Soldati D (2004) The glideosome: a molecular machine powering motility and host-cell invasion by Apicomplexa. Trends Cell Biol 14: 528–532. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

