Aberrant glycogen synthase kinase 3β is involved in pancreatic cancer cell invasion and resistance to therapy
- PMID: 23408967
- PMCID: PMC3568118
- DOI: 10.1371/journal.pone.0055289
Aberrant glycogen synthase kinase 3β is involved in pancreatic cancer cell invasion and resistance to therapy
Abstract
Background and purpose: The major obstacles to treatment of pancreatic cancer are the highly invasive capacity and resistance to chemo- and radiotherapy. Glycogen synthase kinase 3β (GSK3β) regulates multiple cellular pathways and is implicated in various diseases including cancer. Here we investigate a pathological role for GSK3β in the invasive and treatment resistant phenotype of pancreatic cancer.
Methods: Pancreatic cancer cells were examined for GSK3β expression, phosphorylation and activity using Western blotting and in vitro kinase assay. The effects of GSK3β inhibition on cancer cell survival, proliferation, invasive ability and susceptibility to gemcitabine and radiation were examined following treatment with a pharmacological inhibitor or by RNA interference. Effects of GSK3β inhibition on cancer cell xenografts were also examined.
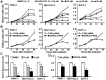
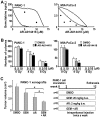
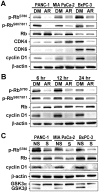
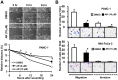
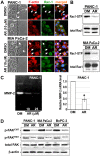
Results: Pancreatic cancer cells showed higher expression and activity of GSK3β than non-neoplastic cells, which were associated with changes in its differential phosphorylation. Inhibition of GSK3β significantly reduced the proliferation and survival of cancer cells, sensitized them to gemcitabine and ionizing radiation, and attenuated their migration and invasion. These effects were associated with decreases in cyclin D1 expression and Rb phosphorylation. Inhibition of GSK3β also altered the subcellular localization of Rac1 and F-actin and the cellular microarchitecture, including lamellipodia. Coincident with these changes were the reduced secretion of matrix metalloproteinase-2 (MMP-2) and decreased phosphorylation of focal adhesion kinase (FAK). The effects of GSK3β inhibition on tumor invasion, susceptibility to gemcitabine, MMP-2 expression and FAK phosphorylation were observed in tumor xenografts.
Conclusion: The targeting of GSK3β represents an effective strategy to overcome the dual challenges of invasiveness and treatment resistance in pancreatic cancer.
Conflict of interest statement
Figures


References
-
- Siegel R, Naishadham D, Jemal A (2012) Cancer statistics, 2012. CA Cancer J Clin 62: 10–29. - PubMed
-
- Bardeesy N, DePinho RA (2002) Pancreatic cancer biology and genetics. Nat Rev Cancer 2: 897–909. - PubMed
-
- Li D, Xie K, Wolff R, Abbruzzese JL (2004) Pancreatic cancer. Lancet 363: 1049–1057. - PubMed
-
- Hidalgo M (2010) Pancreatic cancer. N Engl J Med 362: 1605–1617. - PubMed
-
- Furukawa T (2008) Molecular targeting therapy for pancreatic cancer: Current knowledge and perspectives from bench to bedside. J Gastroenterol 43: 905–911. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

