Transplacental exposure to AZT induces adverse neurochemical and behavioral effects in a mouse model: protection by L-acetylcarnitine
- PMID: 23409035
- PMCID: PMC3567094
- DOI: 10.1371/journal.pone.0055753
Transplacental exposure to AZT induces adverse neurochemical and behavioral effects in a mouse model: protection by L-acetylcarnitine
Erratum in
- PLoS One. 2013;8(10). doi: 10.1371/annotation/e675ab9e-a978-4f9d-a575-67a337964790
Abstract
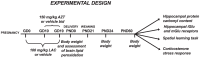
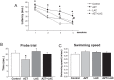
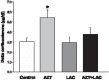
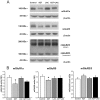
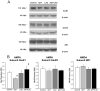
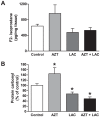
Maternal-fetal HIV-1 transmission can be prevented by administration of AZT, alone or in combination with other antiretroviral drugs to pregnant HIV-1-infected women and their newborns. In spite of the benefits deriving from this life-saving prophylactic therapy, there is still considerable uncertainty on the potential long-term adverse effects of antiretroviral drugs on exposed children. Clinical and experimental studies have consistently shown the occurrence of mitochondrial dysfunction and increased oxidative stress following prenatal treatment with antiretroviral drugs, and clinical evidence suggests that the developing brain is one of the targets of the toxic action of these compounds possibly resulting in behavioral problems. We intended to verify the effects on brain and behavior of mice exposed during gestation to AZT, the backbone of antiretroviral therapy during human pregnancy. We hypothesized that glutamate, a neurotransmitter involved in excitotoxicity and behavioral plasticity, could be one of the major actors in AZT-induced neurochemical and behavioral alterations. We also assessed the antioxidant and neuroprotective effect of L-acetylcarnitine, a compound that improves mitochondrial function and is successfully used to treat antiretroviral-induced polyneuropathy in HIV-1 patients. We found that transplacental exposure to AZT given per os to pregnant mice from day 10 of pregnancy to delivery impaired in the adult offspring spatial learning and memory, enhanced corticosterone release in response to acute stress, increased brain oxidative stress also at birth and markedly reduced expression of mGluR1 and mGluR5 subtypes and GluR1 subunit of AMPA receptors in the hippocampus. Notably, administration during the entire pregnancy of L-acetylcarnitine was effective in preventing/ameliorating the neurochemical, neuroendocrine and behavioral adverse effects induced by AZT in the offspring. The present preclinical findings provide a mechanistic hypothesis for the neurobehavioral effects of AZT and strongly suggest that preventive administration of L-acetylcarnitine might be effective in reducing the neurological side-effects of antiretroviral therapy in fetus/newborn.
Conflict of interest statement
Figures
Similar articles
-
Placental Mitochondrial Toxicity, Oxidative Stress, Apoptosis, and Adverse Perinatal Outcomes in HIV Pregnancies Under Antiretroviral Treatment Containing Zidovudine.J Acquir Immune Defic Syndr. 2017 Aug 1;75(4):e113-e119. doi: 10.1097/QAI.0000000000001334. J Acquir Immune Defic Syndr. 2017. PMID: 28234688
-
Effects of prenatal AZT on mouse neurobehavioral development and passive avoidance learning.Neurotoxicol Teratol. 1999 Jan-Feb;21(1):29-40. doi: 10.1016/s0892-0362(98)00035-x. Neurotoxicol Teratol. 1999. PMID: 10023799
-
Transplacentally exposed human and monkey newborn infants show similar evidence of nucleoside reverse transcriptase inhibitor-induced mitochondrial toxicity.Environ Mol Mutagen. 2007 Apr-May;48(3-4):201-9. doi: 10.1002/em.20201. Environ Mol Mutagen. 2007. PMID: 16538687
-
Animal models of anti-HIV drugs exposure during pregnancy: effects on neurobehavioral development.Prog Neuropsychopharmacol Biol Psychiatry. 2002 May;26(4):747-61. doi: 10.1016/s0278-5846(01)00325-6. Prog Neuropsychopharmacol Biol Psychiatry. 2002. PMID: 12188107 Review.
-
Relevance of experimental models for investigation of genotoxicity induced by antiretroviral therapy during human pregnancy.Mutat Res. 2008 Mar-Apr;658(3):184-90. doi: 10.1016/j.mrrev.2007.12.001. Epub 2008 Jan 12. Mutat Res. 2008. PMID: 18295533 Free PMC article. Review.
Cited by
-
In utero exposure to HIV and/or antiretroviral therapy: a systematic review of preclinical and clinical evidence of cognitive outcomes.J Int AIDS Soc. 2019 Apr;22(4):e25275. doi: 10.1002/jia2.25275. J Int AIDS Soc. 2019. PMID: 30983111 Free PMC article.
-
High-throughput chinmedomics-based prediction of effective components and targets from herbal medicine AS1350.Sci Rep. 2016 Dec 2;6:38437. doi: 10.1038/srep38437. Sci Rep. 2016. PMID: 27910928 Free PMC article.
-
Mitochondrial Toxicogenomics for Antiretroviral Management: HIV Post-exposure Prophylaxis in Uninfected Patients.Front Genet. 2020 May 26;11:497. doi: 10.3389/fgene.2020.00497. eCollection 2020. Front Genet. 2020. PMID: 32528527 Free PMC article.
-
Effects of Zidovudine Treatment on Heart mRNA Expression and Mitochondrial DNA Copy Number Associated with Alterations in Deoxynucleoside Triphosphate Composition in a Neonatal Rat Model.Antimicrob Agents Chemother. 2015 Oct;59(10):6328-36. doi: 10.1128/AAC.01180-15. Epub 2015 Jul 27. Antimicrob Agents Chemother. 2015. PMID: 26248377 Free PMC article.
-
The PHACS SMARTT Study: Assessment of the Safety of In Utero Exposure to Antiretroviral Drugs.Front Immunol. 2016 May 23;7:199. doi: 10.3389/fimmu.2016.00199. eCollection 2016. Front Immunol. 2016. PMID: 27242802 Free PMC article. Review.
References
-
- Mofenson LM (2003) Advances in the prevention of vertical transmission of human immunodeficiency virus. Semin Pediatr Infect Dis 14: 295–308. - PubMed
-
- Thorne C, Newell ML (2007) Safety of agents used to prevent mother-to-child transmission of HIV: is there any cause for concern? Drug Saf 30: 203–213. - PubMed
-
- Cooper ER, Charurat M, Mofenson L, Hanson IC, Pitt J, et al. (2002) Combination antiretroviral strategies for the treatment of pregnant HIV-1-infected women and prevention of perinatal HIV-1 transmission. J Acquir Immune Defic Syndr 29: 484–494. - PubMed
-
- European Collaborative Study (2003) Exposure to antiretroviral therapy in utero or early life: the health of uninfected children born to HIV-infected women. J Acquir Immune Defic Syndr 32: 380–387. - PubMed
-
- Kakuda TN (2000) Pharmacology of nucleoside and nucleotide reverse transcriptase inhibitor-induced mitochondrial toxicity. Clin Ther 22: 685–708. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

