Novel transcriptional regulation of VEGF in inflammatory processes
- PMID: 23414097
- PMCID: PMC3612137
- DOI: 10.1111/jcmm.12020
Novel transcriptional regulation of VEGF in inflammatory processes
Abstract
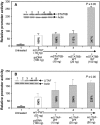
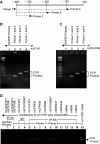
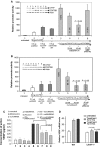
Vascular endothelial growth factor (VEGF) is a critical angiogenic factor affecting endothelial cells, inflammatory cells and neuronal cells. In addition to its well-defined positive role in wound healing, pathological roles for VEGF have been described in cancer and inflammatory diseases (i.e. atherosclerosis, rheumatoid arthritis, inflammatory bowel disease and osteoarthritis). Recently, we showed that transcription factors LITAF and STAT6B affected the inflammatory response. This study builds upon our previous results in testing the role of mouse LITAF and STAT6B in the regulation of VEGF-mediated processes. Cells cotransfected with a series of VEGF promoter deletions along with truncated forms of mLITAF and/or mSTAT6B identified a DNA binding site (between -338 and -305 upstream of the transcription site) important in LITAF and/or STAT6B-mediated transcriptional regulation of VEGF. LITAF and STAT6B corresponding protein sites were identified. In addition, siRNA-mediated knockdown of mLITAF and/or mSTAT6B leads to significant reduction in VEGF mRNA levels and inhibits LPS-induced VEGF secretion in mouse RAW 264.7 cells. Furthermore, VEGF treatment of mouse macrophage or endothelial cells induces LITAF/STAT6B nuclear translocation and cell migration. To translate these observations in vivo, VEGF164-soaked matrigel were implanted in whole-body LITAF-deficient animals (TamLITAF(-/-) ), wild-type mice silenced for STAT6B, and in respective control animals. Vessel formation was found significantly reduced in TamLITAF(-/-) as well as in STAT6B-silenced wild-type animals compared with control animals. The present data demonstrate that VEGF regulation by LITAF and/or STAT6B is important in angiogenesis signalling pathways and may be a useful target in the treatment of VEGF diseases.
© 2013 The Authors. Published by Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures



Similar articles
-
Novel regulation of CCL2 gene expression by murine LITAF and STAT6B.PLoS One. 2011;6(9):e25083. doi: 10.1371/journal.pone.0025083. Epub 2011 Sep 28. PLoS One. 2011. PMID: 21980379 Free PMC article.
-
Crosstalk between VEGF and MTA1 signaling pathways contribute to aggressiveness of breast carcinoma.Mol Carcinog. 2015 May;54(5):333-50. doi: 10.1002/mc.22104. Epub 2013 Nov 22. Mol Carcinog. 2015. PMID: 24265228
-
A natural anti-inflammatory enone fatty acid inhibits angiogenesis by attenuating nuclear factor-κB signaling in vascular endothelial cells.Int J Oncol. 2011 Feb;38(2):493-501. doi: 10.3892/ijo.2010.856. Epub 2010 Dec 3. Int J Oncol. 2011. PMID: 21132269
-
Growing and shaping the vascular tree: multiple roles for VEGF.Bioessays. 2003 Nov;25(11):1052-60. doi: 10.1002/bies.10351. Bioessays. 2003. PMID: 14579246 Review.
-
VEGF-A mRNA processing, stability and translation: a paradigm for intricate regulation of gene expression at the post-transcriptional level.Nucleic Acids Res. 2013 Sep;41(17):7997-8010. doi: 10.1093/nar/gkt539. Epub 2013 Jul 12. Nucleic Acids Res. 2013. PMID: 23851566 Free PMC article. Review.
Cited by
-
Relationship between VEGF Gene Polymorphisms and Serum VEGF Protein Levels in Patients with Rheumatoid Arthritis.PLoS One. 2016 Aug 11;11(8):e0160769. doi: 10.1371/journal.pone.0160769. eCollection 2016. PLoS One. 2016. PMID: 27513931 Free PMC article. Clinical Trial.
-
Discovery of Novel Small Molecule Inhibitors of VEGF Expression in Tumor Cells Using a Cell-Based High Throughput Screening Platform.PLoS One. 2016 Dec 16;11(12):e0168366. doi: 10.1371/journal.pone.0168366. eCollection 2016. PLoS One. 2016. PMID: 27992500 Free PMC article.
-
STAT6 Upregulates NRP1 Expression in Endothelial Cells and Promotes Angiogenesis.Front Oncol. 2022 May 5;12:823377. doi: 10.3389/fonc.2022.823377. eCollection 2022. Front Oncol. 2022. PMID: 35600336 Free PMC article.
-
Prognostic impact of the pretreatment albumin to alkaline phosphatase ratio for nonmetastatic breast cancer patients.Cancer Manag Res. 2019 May 27;11:4809-4814. doi: 10.2147/CMAR.S200759. eCollection 2019. Cancer Manag Res. 2019. PMID: 31213902 Free PMC article.
-
Berberine ameliorates collagen-induced arthritis in rats associated with anti-inflammatory and anti-angiogenic effects.Inflammation. 2014 Oct;37(5):1789-98. doi: 10.1007/s10753-014-9909-y. Inflammation. 2014. PMID: 24803296
References
-
- Peters M, Kauth M, Scherner O, et al. Arabinogalactan isolated from cowshed dust extract protects mice from allergic airway inflammation and sensitization. J Allergy Clin Immunol. 2010;126:648–56. - PubMed
-
- Chen YF, Jobanputra P, Barton P, et al. A systematic review of the effectiveness of adalimumab, etanercept and infliximab for the treatment of rheumatoid arthritis in adults and an economic evaluation of their cost-effectiveness. Health Technol Assess. 2006;10:1–229. - PubMed
-
- Aggarwal BB, Vijayalekshmi RV, Sung B. Targeting inflammatory pathways for prevention and therapy of cancer: short-term friend, long-term foe. Clin Cancer Res. 2009;15:425–30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

