Luminal Ca2+ depletion during the unfolded protein response in Xenopus oocytes: cause and consequence
- PMID: 23415071
- PMCID: PMC3594557
- DOI: 10.1016/j.ceca.2013.01.002
Luminal Ca2+ depletion during the unfolded protein response in Xenopus oocytes: cause and consequence
Abstract
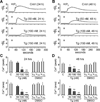
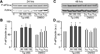
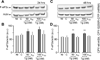
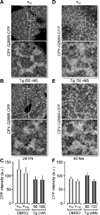
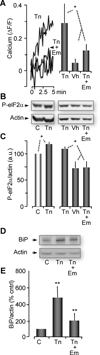
The endoplasmic reticulum (ER) is a Ca(2+) storing organelle that plays a critical role in the synthesis, folding and post-translational modifications of many proteins. The ER enters into a condition of stress when the load of newly synthesized proteins exceeds its folding and processing capacity. This activates a signal transduction pathway called the unfolded protein response (UPR) that attempts to restore homeostasis. The precise role of ER Ca(2+) in the initiation of the UPR has not been defined. Specifically, it has not been established whether ER Ca(2+) dysregulation is a cause or consequence of ER stress. Here, we report that partial depletion of ER Ca(2+) stores induces a significant induction of the UPR, and leads to the retention of a normally secreted protein Carboxypeptidase Y. Moreover, inhibition of protein glycosylation by tunicamycin rapidly induced an ER Ca(2+) leak into the cytosol. However, blockade of the translocon with emetine inhibited the tunicamycin-induced Ca(2+) release. Furthermore, emetine treatment blocked elF2α phosphorylation and reduced expression of the chaperone BiP. These findings suggest that Ca(2+) may be both a cause and a consequence of ER protein misfolding. Thus, it appears that ER Ca(2+) leak is a significant co-factor for the initiation of the UPR.
Copyright © 2013 Elsevier Ltd. All rights reserved.
Figures

References
-
- Alberts BJ, Lewis A, Raff J, Robberts M, Walter, P K. Garland Science. New York, NY: 2002. Molecular Biology of the Cell.
-
- Hammond C, Helenius A. Quality control in the secretory pathway. Curr Opin Cell Biol. 1995;7:523–529. - PubMed
-
- Berridge MJ. The endoplasmic reticulum: a multifunctional signaling organelle. Cell Calcium. 2002;32:235–249. - PubMed
-
- Meldolesi J, Pozzan T. The endoplasmic reticulum Ca2+ store: a view from the lumen. Trends Biochem Sci. 1998;23:10–14. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

