Pancreatic cancer-associated Cathepsin E as a drug activator
- PMID: 23422726
- PMCID: PMC3638719
- DOI: 10.1016/j.jconrel.2013.02.007
Pancreatic cancer-associated Cathepsin E as a drug activator
Abstract
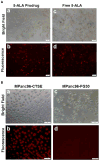
Pancreatic ductal adenocarcinoma (PDAC) is challenging to treat, and better means to detect and/or treat pancreatic cancer are urgently needed to save lives. Cathepsin E (Cath E) is a proteolytic enzyme highly expressed in PDAC. In this study, a novel approach using Cath E activation of a Cath E-specific prodrug was demonstrated. Specific activation of the prodrug is expected to kill pancreatic cancer cells without harming normal pancreatic cells. A novel 5-aminolevulinic acid (5-ALA) prodrug was custom-designed to be activated selectively by endogenous Cath E within the PDAC cells. The 5-ALA prodrug was incubated with Cath E-positive and -negative tumor cells and illuminated with various doses of light. In addition, mice genetically engineered to develop PDAC were injected intravenously with the 5-ALA prodrug, and the pancreas was treated with light irradiation. One day after treatment, PDAC tissue was assessed for apoptosis. The 5-ALA prodrug was activated within the Cath E-positive tumor but not in the normal pancreatic tissue. When used in combination with light treatment, it allowed delivery of selective photodynamic therapy (PDT) to the cancerous tissues, with minimal harm to the adjacent normal tissues. With this novel Cath E activation approach, it is possible to detect pancreatic cancer cells accurately and specifically impair their viability, while sparing normal cells. This treatment could result in fewer side effects than the non-specific treatments currently in use. Cath E is a specific and effective drug activator for PDAC treatment.
Copyright © 2013 Elsevier B.V. All rights reserved.
Figures





References
-
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA: a cancer journal for clinicians. 2012;62(1):10–29. - PubMed
-
- Yeo CJ, Cameron JL, Sohn TA, Lillemoe KD, Pitt HA, Talamini MA, Hruban RH, Ord SE, Sauter PK, Coleman J, Zahurak ML, Grochow LB, Abrams RA. Six hundred fifty consecutive pancreaticoduodenectomies in the 1990s: pathology, complications, and outcomes. Annals of surgery. 1997;226(3):248–257. discussion 257–260. - PMC - PubMed
-
- Brown SB, Brown EA, Walker I. The present and future role of photodynamic therapy in cancer treatment. The lancet oncology. 2004;5(8):497–508. - PubMed
-
- Dolmans DE, Fukumura D, Jain RK. Photodynamic therapy for cancer. Nature reviews. 2003;3(5):380–387. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

