Similar uptake but different trafficking and escape routes of reovirus virions and infectious subvirion particles imaged in polarized Madin-Darby canine kidney cells
- PMID: 23427267
- PMCID: PMC3623640
- DOI: 10.1091/mbc.E12-12-0852
Similar uptake but different trafficking and escape routes of reovirus virions and infectious subvirion particles imaged in polarized Madin-Darby canine kidney cells
Abstract
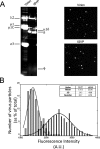
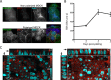
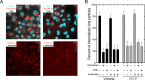
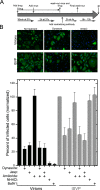
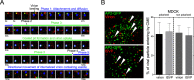
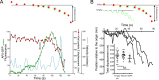
Polarized epithelial cells that line the digestive, respiratory, and genitourinary tracts form a barrier that many viruses must breach to infect their hosts. Current understanding of cell entry by mammalian reovirus (MRV) virions and infectious subvirion particles (ISVPs), generated from MRV virions by extracellular proteolysis in the digestive tract, are mostly derived from in vitro studies with nonpolarized cells. Recent live-cell imaging advances allow us for the first time to visualize events at the apical surface of polarized cells. In this study, we used spinning-disk confocal fluorescence microscopy with high temporal and spatial resolution to follow the uptake and trafficking dynamics of single MRV virions and ISVPs at the apical surface of live polarized Madin-Darby canine kidney cells. Both types of particles were internalized by clathrin-mediated endocytosis, but virions and ISVPs exhibited strikingly different trafficking after uptake. While virions reached early and late endosomes, ISVPs did not and instead escaped the endocytic pathway from an earlier location. This study highlights the broad advantages of using live-cell imaging combined with single-particle tracking for identifying key steps in cell entry by viruses.
Figures


References
-
- Barton ES, Connolly JL, Forrest JC, Chappell JD, Dermody TS. Utilization of sialic acid as a coreceptor enhances reovirus attachment by multistep adhesion strengthening. J Biol Chem. 2001a;276:2200–2211. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

