The mastermind approach to CNS drug therapy: translational prediction of human brain distribution, target site kinetics, and therapeutic effects
- PMID: 23432852
- PMCID: PMC3602026
- DOI: 10.1186/2045-8118-10-12
The mastermind approach to CNS drug therapy: translational prediction of human brain distribution, target site kinetics, and therapeutic effects
Abstract
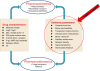
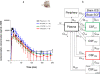
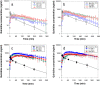
Despite enormous advances in CNS research, CNS disorders remain the world's leading cause of disability. This accounts for more hospitalizations and prolonged care than almost all other diseases combined, and indicates a high unmet need for good CNS drugs and drug therapies.Following dosing, not only the chemical properties of the drug and blood-brain barrier (BBB) transport, but also many other processes will ultimately determine brain target site kinetics and consequently the CNS effects. The rate and extent of all these processes are regulated dynamically, and thus condition dependent. Therefore, heterogenious conditions such as species, gender, genetic background, tissue, age, diet, disease, drug treatment etc., result in considerable inter-individual and intra-individual variation, often encountered in CNS drug therapy.For effective therapy, drugs should access the CNS "at the right place, at the right time, and at the right concentration". To improve CNS therapies and drug development, details of inter-species and inter-condition variations are needed to enable target site pharmacokinetics and associated CNS effects to be translated between species and between disease states. Specifically, such studies need to include information about unbound drug concentrations which drive the effects. To date the only technique that can obtain unbound drug concentrations in brain is microdialysis. This (minimally) invasive technique cannot be readily applied to humans, and we need to rely on translational approaches to predict human brain distribution, target site kinetics, and therapeutic effects of CNS drugs.In this review the term "Mastermind approach" is introduced, for strategic and systematic CNS drug research using advanced preclinical experimental designs and mathematical modeling. In this way, knowledge can be obtained about the contributions and variability of individual processes on the causal path between drug dosing and CNS effect in animals that can be translated to the human situation. On the basis of a few advanced preclinical microdialysis based investigations it will be shown that the "Mastermind approach" has a high potential for the prediction of human CNS drug effects.
Figures





References
-
- World Health Organization. “Neurological Disorders: Public Health Challenges”. 2007.
-
- Neuwelt EA, Abbott NJ, Abrey L, Banks WA, Blakley B, Davis T, Engelhardt B, Grammas P, Nedergaard M, Nutt J, Pardridge W, Rosenburg GA, Smith Q, Drewes LR. Strategies to advance translational research into brain barriers. Lancet Neurol. 2008;7:84–96. doi: 10.1016/S1474-4422(07)70326-5. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

