Bmi1 enhances tumorigenicity and cancer stem cell function in pancreatic adenocarcinoma
- PMID: 23437065
- PMCID: PMC3577834
- DOI: 10.1371/journal.pone.0055820
Bmi1 enhances tumorigenicity and cancer stem cell function in pancreatic adenocarcinoma
Abstract
Background: Bmi1 is an integral component of the Polycomb Repressive Complex 1 (PRC1) and is involved in the pathogenesis of multiple cancers. It also plays a key role in the functioning of endogenous stem cells and cancer stem cells. Previous work implicated a role for cancer stem cells in the pathogenesis of pancreatic cancer. We hypothesized that Bmi1 plays an integral role in enhancing pancreatic tumorigenicity and the function of cancer stem cells in pancreatic ductal adenocarcinoma.
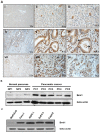
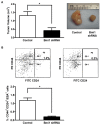
Methods: We measured endogenous Bmi1 levels in primary human pancreatic ductal adenocarcinomas, pancreatic intraepithelial neoplasias (PanINs) and normal pancreas by immunohistochemistry and Western blotting. The function of Bmi1 in pancreatic cancer was assessed by alteration of Bmi1 expression in several cell model systems by measuring cell proliferation, cell apoptosis, in vitro invasion, chemotherapy resistance, and in vivo growth and metastasis in an orthotopic model of pancreatic cancer. We also assessed the cancer stem cell frequency, tumorsphere formation, and in vivo growth of human pancreatic cancer xenografts after Bmi1 silencing.
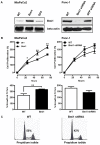
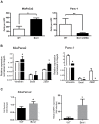
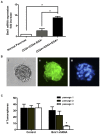
Results: Bmi1 was overexpressed in human PanINs, pancreatic cancers, and in several pancreatic cancer cell lines. Overexpression of Bmi1 in MiaPaCa2 cells resulted in increased proliferation, in vitro invasion, larger in vivo tumors, more metastases, and gemcitabine resistance while opposite results were seen when Bmi1 was silenced in Panc-1 cells. Bmi1 was overexpressed in the cancer stem cell compartment of primary human pancreatic cancer xenografts. Pancreatic tumorspheres also demonstrated high levels of Bmi1. Silencing of Bmi1 inhibited secondary and tertiary tumorsphere formation, decreased primary pancreatic xenograft growth, and lowered the proportion of cancer stem cells in the xenograft tissue.
Conclusions: Our results implicate Bmi1 in the invasiveness and growth of pancreatic cancer and demonstrate its key role in the regulation of pancreatic cancer stem cells.
Conflict of interest statement
Figures

References
-
- SEER website. Available: http://seer.cancer.gov/csr/1975_2009_pops09/results_single/sect_01_table.... Accessed 2013 Jan 1.
-
- Haupt Y, Bath ML, Harris AW, Adams JM (1993) bmi-1 transgene induces lymphomas and collaborates with myc in tumorigenesis. Oncogene 8: 3161–3164. - PubMed
-
- van Lohuizen M, Verbeek S, Scheijen B, Wientjens E, van der Gulden H, et al. (1991) Identification of cooperating oncogenes in E mu-myc transgenic mice by provirus tagging. Cell 65: 737–752. - PubMed
-
- Jacobs JJ, Kieboom K, Marino S, DePinho RA, van Lohuizen M (1999) The oncogene and Polycomb-group gene bmi-1 regulates cell proliferation and senescence through the ink4a locus. Nature 397: 164–168. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

