Calreticulin induces dilated cardiomyopathy
- PMID: 23437120
- PMCID: PMC3577809
- DOI: 10.1371/journal.pone.0056387
Calreticulin induces dilated cardiomyopathy
Erratum in
- PLoS One. 2013;8(11). doi:10.1371/annotation/04c791d5-a71c-493d-b94a-a57b158c5538
Abstract
Background: Calreticulin, a Ca(2+)-buffering chaperone of the endoplasmic reticulum, is highly expressed in the embryonic heart and is essential for cardiac development. After birth, the calreticulin gene is sharply down regulated in the heart, and thus, adult hearts have negligible levels of calreticulin. In this study we tested the role of calreticulin in the adult heart.
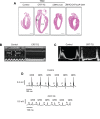
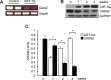
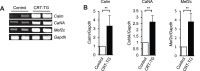
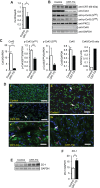
Methodology/principal findings: We generated an inducible transgenic mouse in which calreticulin is targeted to the cardiac tissue using a Cre/loxP system and can be up-regulated in adult hearts. Echocardiography analysis of hearts from transgenic mice expressing calreticulin revealed impaired left ventricular systolic and diastolic function and impaired mitral valve function. There was altered expression of Ca(2+) signaling molecules and the gap junction proteins, Connexin 43 and 45. Sarcoplasmic reticulum associated Ca(2+)-handling proteins (including the cardiac ryanodine receptor, sarco/endoplasmic reticulum Ca(2+)-ATPase, and cardiac calsequestrin) were down-regulated in the transgenic hearts with increased expression of calreticulin.
Conclusions/significance: We show that in adult heart, up-regulated expression of calreticulin induces cardiomyopathy in vivo leading to heart failure. This is due to an alternation in changes in a subset of Ca(2+) handling genes, gap junction components and left ventricle remodeling.
Conflict of interest statement
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

