A motivational interviewing intervention to PREvent PAssive Smoke Exposure (PREPASE) in children with a high risk of asthma: design of a randomised controlled trial
- PMID: 23442389
- PMCID: PMC3599824
- DOI: 10.1186/1471-2458-13-177
A motivational interviewing intervention to PREvent PAssive Smoke Exposure (PREPASE) in children with a high risk of asthma: design of a randomised controlled trial
Abstract
Background: Especially children at risk for asthma are sensitive to the detrimental health effects of passive smoke (PS) exposure, like respiratory complaints and allergic sensitisation. Therefore, effective prevention of PS exposure in this group of vulnerable children is important. Based on previous studies, we hypothesized that an effective intervention program to prevent PS exposure in children is possible by means of a motivational interviewing tailored program with repeated contacts focussing on awareness, knowledge, beliefs (pros/cons), perceived barriers and needs of parents, in combination with feedback about urine cotinine levels of the children. The aim of the PREPASE study is to test the effectiveness of such an intervention program towards eliminating or reducing of PS exposure in children at risk for asthma. This article describes the protocol of the PREPASE study.
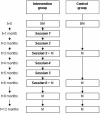
Methods: The study is a one-year follow-up randomized controlled trial. Families with children (0-13 years of age) having an asthma predisposition who experience PS exposure at home are randomized into an intervention group receiving an intervention or a control group receiving care as usual. The intervention is given by trained research assistants. The intervention starts one month after a baseline measurement and takes place once per month for an hour during six home based counselling sessions. The primary outcome measure is the percentage of families curtailing PS exposure in children (parental report verified with the urine cotinine concentrations of the children) after 6 months. The secondary outcome measures include: household nicotine level, the child's lung function, airway inflammation and oxidative stress, presence of wheezing and questionnaires on respiratory symptoms, and quality of life. A process evaluation is included. Most of the measurements take place every 3 months (baseline and after 3, 6, 9 and 12 months of study).
Conclusion: The PREPASE study incorporates successful elements of previous interventions and may therefore be very promising. If proven effective, the intervention will benefit the health of children at risk for asthma and may also create opportunity to be tested in other population.
Trial registration number: NTR2632.
Figures

References
-
- WHO report on the gLobal tobacco epidemic, 2009: implementing smoke-free environments. http://www.who.int/tobacco/mpower/2009/en/index.html.
-
- STIVORO. Key Numbers 2011. http://stivoro.nl/wp-content/uploads/2012/docs/factsheets/Kerncijfers%20.... [cited 2012 October 11.
-
- Crone MR, Nagelhout GE, van den Burg I, HiraSing RA. Passive smoking in young children in the Netherlands sharply decreased since 1996. Ned Tijdschr Geneeskd. 2010;154:A1658. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

