Biophysical model of bacterial cell interactions with nanopatterned cicada wing surfaces
- PMID: 23442962
- PMCID: PMC3576530
- DOI: 10.1016/j.bpj.2012.12.046
Biophysical model of bacterial cell interactions with nanopatterned cicada wing surfaces
Abstract
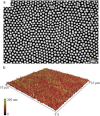
The nanopattern on the surface of Clanger cicada (Psaltoda claripennis) wings represents the first example of a new class of biomaterials that can kill bacteria on contact based solely on their physical surface structure. The wings provide a model for the development of novel functional surfaces that possess an increased resistance to bacterial contamination and infection. We propose a biophysical model of the interactions between bacterial cells and cicada wing surface structures, and show that mechanical properties, in particular cell rigidity, are key factors in determining bacterial resistance/sensitivity to the bactericidal nature of the wing surface. We confirmed this experimentally by decreasing the rigidity of surface-resistant strains through microwave irradiation of the cells, which renders them susceptible to the wing effects. Our findings demonstrate the potential benefits of incorporating cicada wing nanopatterns into the design of antibacterial nanomaterials.
Copyright © 2013 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Marmur A. The Lotus effect: superhydrophobicity and metastability. Langmuir. 2004;20:3517–3519. - PubMed
-
- Su Y., Ji B., Hwang K.C. Nature’s design of hierarchical superhydrophobic surfaces of a water strider for low adhesion and low-energy dissipation. Langmuir. 2010;26:18926–18937. - PubMed
-
- Bhushan B., Jung Y.C. Natural and biomimetic artificial surfaces for superhydrophobicity, self-cleaning, low adhesion, and drag reduction. Prog. Mater. Sci. 2011;56:1–108.
-
- Guo Z., Liu W., Su B.-L. Superhydrophobic surfaces: from natural to biomimetic to functional. J. Colloid Interface Sci. 2011;353:335–355. - PubMed
-
- Webb H.K., Hasan J., Ivanova E.P. Nature inspired structured surfaces for biomedical applications. Curr. Med. Chem. 2011;18:3367–3375. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

