Do clustering monoclonal antibody solutions really have a concentration dependence of viscosity?
- PMID: 23442970
- PMCID: PMC3576527
- DOI: 10.1016/j.bpj.2013.01.007
Do clustering monoclonal antibody solutions really have a concentration dependence of viscosity?
Abstract
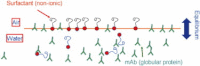
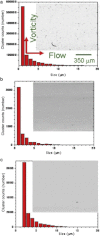
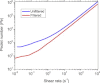
Protein solution rheology data in the biophysics literature have incompletely identified factors that govern hydrodynamics. Whereas spontaneous protein adsorption at the air/water (A/W) interface increases the apparent viscosity of surfactant-free globular protein solutions, it is demonstrated here that irreversible clusters also increase system viscosity in the zero shear limit. Solution rheology measured with double gap geometry in a stress-controlled rheometer on a surfactant-free Immunoglobulin solution demonstrated that both irreversible clusters and the A/W interface increased the apparent low shear rate viscosity. Interfacial shear rheology data showed that the A/W interface yields, i.e., shows solid-like behavior. The A/W interface contribution was smaller, yet nonnegligible, in double gap compared to cone-plate geometry. Apparent nonmonotonic composition dependence of viscosity at low shear rates due to irreversible (nonequilibrium) clusters was resolved by filtration to recover a monotonically increasing viscosity-concentration curve, as expected. Although smaller equilibrium clusters also existed, their size and effective volume fraction were unaffected by filtration, rendering their contribution to viscosity invariant. Surfactant-free antibody systems containing clusters have complex hydrodynamic response, reflecting distinct bulk and interface-adsorbed protein as well as irreversible cluster contributions. Literature models for solution viscosity lack the appropriate physics to describe the bulk shear viscosity of unstable surfactant-free antibody solutions.
Copyright © 2013 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures






References
-
- Köhler G., Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975;256:495–497. - PubMed
-
- The Nobel Prize in Physiology or Medicine. 1984. http://www.nobelprize.org/nobel_prizes/medicine/laureates/1984/.
-
- Leader B., Baca Q.J., Golan D.E. Protein therapeutics: a summary and pharmacological classification. Nat. Rev. Drug Discov. 2008;7:21–39. - PubMed
-
- Shire S.J. Formulation and manufacturability of biologics. Curr. Opin. Biotechnol. 2009;20:708–714. - PubMed
-
- DeYoung L.R., Fink A.L., Dill K.A. Aggregation of globular proteins. Acc. Chem. Res. 1993;26:614–620.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

