Low-molecular-mass metal complexes in the mouse brain
- PMID: 23443205
- PMCID: PMC3608428
- DOI: 10.1039/c3mt00009e
Low-molecular-mass metal complexes in the mouse brain
Abstract
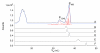
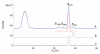
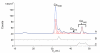
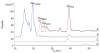
The presence of labile low-molecular-mass (LMM, defined as <10 kDa) metal complexes in cells and super-cellular structures such as the brain has been inferred from chelation studies, but direct evidence is lacking. To evaluate the presence of LMM metal complexes in the brain, supernatant fractions of fresh mouse brain homogenates were passed through a 10 kDa cutoff membrane and subjected to size-exclusion liquid chromatography under anaerobic refrigerated conditions. Fractions were monitored for Mn, Fe, Co, Cu, Zn, Mo, S and P using an on-line ICP-MS. At least 30 different LMM metal complexes were detected along with numerous P- and S- containing species. Reproducibility was assessed by performing the experiment 13 times, using different buffers, and by examining whether complexes changed with time. Eleven Co, 2 Cu, 5 Mn, 4 Mo, 3 Fe and 2 Zn complexes with molecular masses <4 kDa were detected. One LMM Mo complex comigrated with the molybdopterin cofactor. Most Cu and Zn complexes appeared to be protein-bound with masses ranging from 4-20 kDa. Co was the only metal for which the "free" or aqueous complex was reproducibly observed. Aqueous Co may be sufficiently stable in this environment due to its relatively slow water-exchange kinetics. Attempts were made to assign some of these complexes, but further efforts will be required to identify them unambiguously and to determine their functions. This is among the first studies to detect low-molecular-mass transition metal complexes in the mouse brain using LC-ICP-MS.
Figures




References
-
- Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M. Chem-Biol Interact. 2006;160:1–40. - PubMed
-
- Galaris D, Pantopoulos K. Crit Rev Cl Lab Sci. 2008;45:1–23. - PubMed
-
- Ba LA, Doering M, Burkholz T, Jacob C. Metallomics. 2009;1:292–311. - PubMed
-
- Crichton RR, Wilmet S, Legssyer R, Ward RJ. J Inorg Biochem. 2002;91:9–18. - PubMed
-
- Moos T, Morgan EH. Ann Ny Acad Sci. 2004;1012:14–26. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

