A role for Rab14 in the endocytic trafficking of GLUT4 in 3T3-L1 adipocytes
- PMID: 23444368
- PMCID: PMC3666250
- DOI: 10.1242/jcs.104307
A role for Rab14 in the endocytic trafficking of GLUT4 in 3T3-L1 adipocytes
Abstract
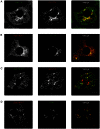
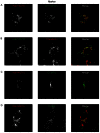
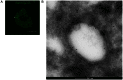
Insulin enhances the uptake of glucose into adipocytes and muscle cells by promoting the redistribution of the glucose transporter isoform 4 (GLUT4) from intracellular compartments to the cell surface. Rab GTPases regulate the trafficking itinerary of GLUT4 and several have been found on immunopurified GLUT4 vesicles. Specifically, Rab14 has previously been implicated in GLUT4 trafficking in muscle although its role, if any, in adipocytes is poorly understood. Analysis of 3T3-L1 adipocytes using confocal microscopy demonstrated that endogenous GLUT4 and endogenous Rab14 exhibited a partial colocalisation. However, when wild-type Rab14 or a constitutively-active Rab14Q70L mutant were overexpressed in these cells, the colocalisation with both GLUT4 and IRAP became extensive. Interestingly, this colocalisation was restricted to enlarged 'ring-like' vesicular structures (mean diameter 1.3 µm), which were observed in the presence of overexpressed wild-type Rab14 and Rab14Q70L, but not an inactive Rab14S25N mutant. These enlarged vesicles contained markers of early endosomes and were rapidly filled by GLUT4 and transferrin undergoing endocytosis from the plasma membrane. The Rab14Q70L mutant reduced basal and insulin-stimulated cell surface GLUT4 levels, probably by retaining GLUT4 in an insulin-insensitive early endosomal compartment. Furthermore, shRNA-mediated depletion of Rab14 inhibited the transit of GLUT4 through early endosomal compartments towards vesicles and tubules in the perinuclear region. Given the previously reported role of Rab14 in trafficking between endosomes and the Golgi complex, we propose that the primary role of Rab14 in GLUT4 trafficking is to control the transit of internalised GLUT4 from early endosomes into the Golgi complex, rather than direct GLUT4 translocation to the plasma membrane.
Keywords: Adipocyte; GLUT4; Insulin; Intracellular trafficking; Rab14.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

