Global, multicenter, randomized, phase II trial of gemcitabine and gemcitabine plus AGS-1C4D4 in patients with previously untreated, metastatic pancreatic cancer
- PMID: 23448807
- PMCID: PMC3716216
- DOI: 10.1093/annonc/mdt066
Global, multicenter, randomized, phase II trial of gemcitabine and gemcitabine plus AGS-1C4D4 in patients with previously untreated, metastatic pancreatic cancer
Abstract
Background: We evaluated AGS-1C4D4, a fully human monoclonal antibody to prostate stem cell antigen (PSCA), with gemcitabine in a randomized, phase II study of metastatic pancreatic cancer.
Patients and methods: Patients with Eastern Cooperative Oncology Group (ECOG) performance status 0/1 and previously untreated, metastatic pancreatic adenocarcinoma were randomly assigned 1:2 to gemcitabine (1000 mg/m(2) weekly seven times, 1 week rest, weekly three times q4weeks) or gemcitabine plus AGS-1C4D4 (48 mg/kg loading dose, then 24 mg/kg q3weeks IV). The primary end point was 6-month survival rate (SR). Archived tumor samples were collected for pre-planned analyses by PSCA expression.
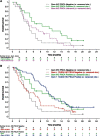
Results: Between April 2009 and May 2010, 196 patients were randomly assigned to gemcitabine (n = 63) or gemcitabine plus AGS-1C4D4 (n = 133). The 6-month SR was 44.4% (95% CI, 31.9-57.5) in the gemcitabine arm and 60.9% (95% CI, 52.1-69.2) in the gemcitabine plus AGS-1C4D4 arm (P = 0.03), while the median survival was 5.5 versus 7.6 months and the response rate was 13.1% versus 21.6% in the two arms, respectively. The 6-month SR was 57.1% in the gemcitabine arm versus 79.5% in the gemcitabine plus AGS-1C4D4 arm among the PSCA-positive subgroup and 31.6% versus 46.2% among the PSCA-negative subgroup.
Conclusions: This randomized, phase II study achieved its primary end point, demonstrating an improved 6-month SR with addition of AGS-1C4D4 to gemcitabine among patients with previously untreated, metastatic pancreatic adenocarcinoma. ClinicalTrials.gov identifier: NCT00902291.
Keywords: chemotherapy; clinical trial; gemcitabine; metastatic disease; pancreatic cancer; prostate stem cell antigen.
Figures

References
-
- Siegel R, Ward E, Brawley O, et al. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61:212–236. - PubMed
-
- Burris HA, III, Moore MJ, Andersen J, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol. 1997;15:2403–2413. - PubMed
-
- Berlin JD, Catalano P, Thomas JP, et al. Phase III study of gemcitabine in combination with fluorouracil versus gemcitabine alone in patients with advanced pancreatic carcinoma: Eastern Cooperative Oncology Group Trial E2297. J Clin Oncol. 2002;20:3270–3275. - PubMed
-
- Rocha Lima CM, Green MR, Rotche R, et al. Irinotecan plus gemcitabine results in no survival advantage compared with gemcitabine monotherapy in patients with locally advanced or metastatic pancreatic cancer despite increased tumor response rate. J Clin Oncol. 2004;22:3776–3783. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

