Combined in vivo depletion of glycoprotein VI and C-type lectin-like receptor 2 severely compromises hemostasis and abrogates arterial thrombosis in mice
- PMID: 23448972
- PMCID: PMC6485540
- DOI: 10.1161/ATVBAHA.112.300672
Combined in vivo depletion of glycoprotein VI and C-type lectin-like receptor 2 severely compromises hemostasis and abrogates arterial thrombosis in mice
Abstract
Objective: Platelet inhibition is a major strategy to prevent acute ischemic cardiovascular and cerebrovascular events, which may, however, be associated with an increased bleeding risk. The (hem)immunoreceptor tyrosine activation motif-bearing platelet receptors, glycoprotein VI (GPVI) and C-type lectin-like receptor 2 (CLEC-2), might be promising antithrombotic targets because they can be depleted from circulating platelets by antibody treatment, leading to sustained antithrombotic protection, but only moderately increased bleeding times in mice.
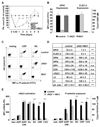
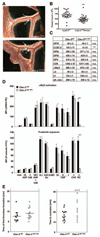
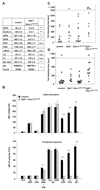
Approach and results: We investigated whether both (hem)immunoreceptor tyrosine activation motif-bearing receptors can be targeted simultaneously and what the in vivo consequences of such a combined therapeutic GPVI/CLEC-2 deficiency are. We demonstrate that isolated targeting of either GPVI or CLEC-2 in vivo does not affect expression or function of the respective other receptor. Moreover, simultaneous treatment with both antibodies resulted in the sustained loss of both GPVI and CLEC-2, while leaving other activation pathways intact. However, GPVI/CLEC-2-depleted mice displayed a dramatic hemostatic defect and profound impairment of arterial thrombus formation. Furthermore, a strongly diminished hemostatic response could also be reproduced in mice genetically lacking GPVI and CLEC-2.
Conclusions: These results demonstrate that GPVI and CLEC-2 can be simultaneously downregulated in platelets in vivo and reveal an unexpected functional redundancy of the 2 receptors in hemostasis and thrombosis. These findings may have important implications of the potential use of anti-GPVI and anti-CLEC-2-based agents in the prevention of thrombotic diseases.
Figures


Comment in
-
Platelet hem-immunoreceptor tyrosine-based activation motif receptors: at least one required.Arterioscler Thromb Vasc Biol. 2013 May;33(5):884-5. doi: 10.1161/ATVBAHA.113.301400. Arterioscler Thromb Vasc Biol. 2013. PMID: 23576714 No abstract available.
Similar articles
-
Galectin-9 activates platelet ITAM receptors glycoprotein VI and C-type lectin-like receptor-2.J Thromb Haemost. 2022 Apr;20(4):936-950. doi: 10.1111/jth.15625. Epub 2022 Jan 6. J Thromb Haemost. 2022. PMID: 34936188
-
Targeting of C-type lectin-like receptor 2 or P2Y12 for the prevention of platelet activation by immunotherapeutic CpG oligodeoxynucleotides.J Thromb Haemost. 2017 May;15(5):983-997. doi: 10.1111/jth.13669. Epub 2017 Apr 13. J Thromb Haemost. 2017. PMID: 28296036
-
Platelet glycoprotein VI and C-type lectin-like receptor 2 deficiency accelerates wound healing by impairing vascular integrity in mice.Haematologica. 2019 Aug;104(8):1648-1660. doi: 10.3324/haematol.2018.208363. Epub 2019 Feb 7. Haematologica. 2019. PMID: 30733265 Free PMC article.
-
GPVI and CLEC-2 in hemostasis and vascular integrity.J Thromb Haemost. 2010 Jul;8(7):1456-67. doi: 10.1111/j.1538-7836.2010.03875.x. Epub 2010 Mar 25. J Thromb Haemost. 2010. PMID: 20345705 Review.
-
Novel antiplatelet strategies targeting GPVI, CLEC-2 and tyrosine kinases.Platelets. 2021 Jan 2;32(1):29-41. doi: 10.1080/09537104.2020.1849600. Epub 2020 Dec 13. Platelets. 2021. PMID: 33307909 Review.
Cited by
-
Glycoprotein VI in securing vascular integrity in inflamed vessels.Res Pract Thromb Haemost. 2018 Apr 3;2(2):228-239. doi: 10.1002/rth2.12092. eCollection 2018 Apr. Res Pract Thromb Haemost. 2018. PMID: 30046725 Free PMC article.
-
Dual-specificity phosphatase 3 deficiency or inhibition limits platelet activation and arterial thrombosis.Circulation. 2015 Feb 17;131(7):656-68. doi: 10.1161/CIRCULATIONAHA.114.010186. Epub 2014 Dec 17. Circulation. 2015. PMID: 25520375 Free PMC article.
-
Targeted downregulation of platelet CLEC-2 occurs through Syk-independent internalization.Blood. 2015 Jun 25;125(26):4069-77. doi: 10.1182/blood-2014-11-611905. Epub 2015 Mar 20. Blood. 2015. PMID: 25795918 Free PMC article.
-
Anti-miR-148a regulates platelet FcγRIIA signaling and decreases thrombosis in vivo in mice.Blood. 2015 Dec 24;126(26):2871-81. doi: 10.1182/blood-2015-02-631135. Epub 2015 Oct 29. Blood. 2015. PMID: 26516227 Free PMC article.
-
Platelet-Cancer Interplay: Molecular Mechanisms and New Therapeutic Avenues.Front Oncol. 2021 Jul 12;11:665534. doi: 10.3389/fonc.2021.665534. eCollection 2021. Front Oncol. 2021. PMID: 34322381 Free PMC article. Review.
References
-
- Michelson AD. Antiplatelet therapies for the treatment of cardiovascular disease. Nat Rev Drug Discov. 2010;9:154–169. - PubMed
-
- Jackson SP. Arterial thrombosis–insidious, unpredictable and deadly. Nat Med. 2011;17:1423–1436. - PubMed
-
- Nieswandt B, Pleines I, Bender M. Platelet adhesion and activation mechanisms in arterial thrombosis and ischaemic stroke. J Thromb Haemost. 2011;9(suppl 1):92–104. - PubMed
-
- Arthur JF, Dunkley S, Andrews RK. Platelet glycoprotein VI-related clinical defects. Br J Haematol. 2007;139:363–372. - PubMed
-
- Kato K, Kanaji T, Russell S, Kunicki TJ, Furihata K, Kanaji S, Marchese P, Reininger A, Ruggeri ZM, Ware J. The contribution of glycoprotein VI to stable platelet adhesion and thrombus formation illustrated by targeted gene deletion. Blood. 2003;102:1701–1707. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

