Effects of interleukin-1 blockade with anakinra on adverse cardiac remodeling and heart failure after acute myocardial infarction [from the Virginia Commonwealth University-Anakinra Remodeling Trial (2) (VCU-ART2) pilot study]
- PMID: 23453459
- PMCID: PMC3644511
- DOI: 10.1016/j.amjcard.2013.01.287
Effects of interleukin-1 blockade with anakinra on adverse cardiac remodeling and heart failure after acute myocardial infarction [from the Virginia Commonwealth University-Anakinra Remodeling Trial (2) (VCU-ART2) pilot study]
Abstract
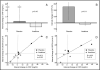
A first pilot study of interleukin-1 blockade in ST-segment elevation acute myocardial infarction showed improved remodeling. In the present second pilot study, we enrolled 30 patients with clinically stable ST-segment elevation acute myocardial infarction randomized to anakinra, recombinant interleukin-1 receptor antagonist, 100 mg/day for 14 days or placebo in a double-blind fashion. The primary end point was the difference in the interval change in left ventricular (LV) end-systolic volume index between the 2 groups within 10 to 14 weeks. The secondary end points included changes in the LV end-diastolic volume index, LV ejection fraction, and C-reactive protein levels. No significant changes in end-systolic volume index, LV end-diastolic volume index, or LV ejection fraction were seen in the placebo group. Compared to placebo, treatment with anakinra led to no measurable differences in these parameters. Anakinra significantly blunted the increase in C-reactive protein between admission and 72 hours (+0.8 mg/dl, interquartile range -6.4 to +4.2, vs +21.1 mg/dl, interquartile range +8.7 to +36.6, p = 0.002), which correlated with the changes in LV end-diastolic volume index and LV end-systolic volume index at 10 to 14 weeks (R = +0.83, p = 0.002, and R = +0.55, p = 0.077, respectively). One patient in the placebo group (7%) died. One patient (7%) in the anakinra group developed recurrent acute myocardial infarction. More patients were diagnosed with new-onset heart failure in the placebo group (4, 27%) than in the anakinra group (1, 7%; p = 0.13). When the data were pooled with those from the first Virginia Commonwealth University-Anakinra Remodeling Trial (n = 40), this difference reached statistical significance (30% vs 5%, p = 0.035). In conclusion, interleukin-1 blockade with anakinra blunted the acute inflammatory response associated with ST-segment elevation acute myocardial infarction. Although it failed to show a statistically significant effect on LV end-systolic volume index, LV end-diastolic volume index, or LV ejection fraction in this cohort of clinically stable patients with near-normal LV dimensions and function, anakinra led to a numerically lower incidence of heart failure.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures



References
-
- Goldberg R, Spencer F, Yarzebski J, Lessard D, Gore J, Alpert J, Dalen JA. 25-Year perspective into the changing landscape of patients hospitalized with acute myocardial infarction (the Worcester Heart Attack Study). Am J Cardiol. 2004;94:1373–1378. - PubMed
-
- Dinarello C. Immunological and inflammatory functions of the interleukin-1 family. Annu Rev Immunol. 2009;27:519–550. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

