Antigenicity and immunogenicity of Plasmodium vivax merozoite surface protein-3
- PMID: 23457498
- PMCID: PMC3573074
- DOI: 10.1371/journal.pone.0056061
Antigenicity and immunogenicity of Plasmodium vivax merozoite surface protein-3
Abstract
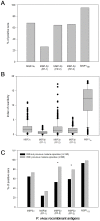
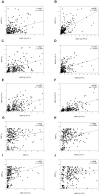
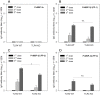
A recent clinical trial in African children demonstrated the potential utility of merozoite surface protein (MSP)-3 as a vaccine against Plasmodium falciparum malaria. The present study evaluated the use of Plasmodium vivax MSP-3 (PvMSP-3) as a target antigen in vaccine formulations against malaria caused by P. vivax. Recombinant proteins representing MSP-3α and MSP-3β of P. vivax were expressed as soluble histidine-tagged bacterial fusions. Antigenicity during natural infection was evaluated by detecting specific antibodies using sera from individuals living in endemic areas of Brazil. A large proportion of infected individuals presented IgG antibodies to PvMSP-3α (68.2%) and at least 1 recombinant protein representing PvMSP-3β (79.1%). In spite of the large responder frequency, reactivity to both antigens was significantly lower than was observed for the immunodominant epitope present on the 19-kDa C-terminal region of PvMSP-1. Immunogenicity of the recombinant proteins was studied in mice in the absence or presence of different adjuvant formulations. PvMSP-3β, but not PvMSP-3α, induced a TLR4-independent humoral immune response in the absence of any adjuvant formulation. The immunogenicity of the recombinant antigens were also tested in formulations containing different adjuvants (Alum, Salmonella enterica flagellin, CpG, Quil A,TiterMax® and incomplete Freunds adjuvant) and combinations of two adjuvants (Alum plus flagellin, and CpG plus flagellin). Recombinant PvMSP-3α and PvMSP-3β elicited higher antibody titers capable of recognizing P. vivax-infected erythrocytes harvested from malaria patients. Our results confirm that P. vivax MSP-3 antigens are immunogenic during natural infection, and the corresponding recombinant proteins may be useful in elucidating their vaccine potential.
Conflict of interest statement
Figures




Similar articles
-
Immunogenicity and antigenicity of Plasmodium vivax merozoite surface protein 10.Parasitol Res. 2014 Jul;113(7):2559-68. doi: 10.1007/s00436-014-3907-8. Epub 2014 Apr 25. Parasitol Res. 2014. PMID: 24764159
-
Invasion-inhibitory antibodies elicited by immunization with Plasmodium vivax apical membrane antigen-1 expressed in Pichia pastoris yeast.Infect Immun. 2014 Mar;82(3):1296-307. doi: 10.1128/IAI.01169-13. Epub 2013 Dec 30. Infect Immun. 2014. PMID: 24379279 Free PMC article.
-
Comparison of immunogenicities of recombinant Plasmodium vivax merozoite surface protein 1 19- and 42-kiloDalton fragments expressed in Escherichia coli.Infect Immun. 2004 Oct;72(10):5775-82. doi: 10.1128/IAI.72.10.5775-5782.2004. Infect Immun. 2004. PMID: 15385477 Free PMC article.
-
Plasmodium vivax malaria vaccine development.Mol Immunol. 2001 Dec;38(6):443-55. doi: 10.1016/s0161-5890(01)00080-3. Mol Immunol. 2001. PMID: 11741694 Review.
-
Guidance on the evaluation of Plasmodium vivax vaccines in populations exposed to natural infection.Vaccine. 2009 Sep 18;27(41):5633-43. doi: 10.1016/j.vaccine.2009.07.018. Epub 2009 Jul 26. Vaccine. 2009. PMID: 19638270 Free PMC article. Review.
Cited by
-
Immunogenicity of a Bivalent Adjuvanted Glycoconjugate Vaccine against Salmonella Typhimurium and Salmonella Enteritidis.Front Immunol. 2017 Feb 27;8:168. doi: 10.3389/fimmu.2017.00168. eCollection 2017. Front Immunol. 2017. PMID: 28289411 Free PMC article.
-
CD4+ T cell response correlates with naturally acquired antibodies against Plasmodium vivax tryptophan-rich antigens.Infect Immun. 2015 May;83(5):2018-29. doi: 10.1128/IAI.03095-14. Epub 2015 Mar 2. Infect Immun. 2015. PMID: 25733522 Free PMC article.
-
Anti-Plasmodium vivax merozoite surface protein 3 ϒ (PvMSP3 ϒ) antibodies upon natural infection.Sci Rep. 2024 Apr 26;14(1):9595. doi: 10.1038/s41598-024-59153-w. Sci Rep. 2024. PMID: 38671033 Free PMC article.
-
Naturally acquired immune responses to P. vivax merozoite surface protein 3α and merozoite surface protein 9 are associated with reduced risk of P. vivax malaria in young Papua New Guinean children.PLoS Negl Trop Dis. 2013 Nov 14;7(11):e2498. doi: 10.1371/journal.pntd.0002498. eCollection 2013 Nov. PLoS Negl Trop Dis. 2013. PMID: 24244763 Free PMC article.
-
Merozoite surface protein-3 alpha as a genetic marker for epidemiologic studies in Plasmodium vivax: a cautionary note.Malar J. 2013 Aug 21;12:288. doi: 10.1186/1475-2875-12-288. Malar J. 2013. PMID: 23964962 Free PMC article.
References
-
- Agnandji ST, Lell B, Soulanoudjingar SS, Fernandes JF, Abossolo BP, et al. (2011) First Results of Phase 3 Trial of RTS,S/AS01 Malaria Vaccine in African Children. N Engl J Med 365: 1863–1875. - PubMed
-
- Sirima SB, Cousens S, Druilhe P (2011) Protection against malaria by MSP3 candidate vaccine. N Engl J Med 365: 1062–1064. - PubMed
-
- Arévalo-Herrera M, Soto L, Perlaza BL, Céspedes N, Vera O, et al.. (2011) Antibody-mediated and cellular immune responses induced in naive volunteers by vaccination with long synthetic peptides derived from the Plasmodium vivax circumsporozoite protein. Am J Trop Med Hyg 84(2 Suppl): 35–42. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

