Single Ca2+ channels and exocytosis at sensory synapses
- PMID: 23459757
- PMCID: PMC3717220
- DOI: 10.1113/jphysiol.2012.249482
Single Ca2+ channels and exocytosis at sensory synapses
Abstract
Hair cell synapses in the ear and photoreceptor synapses in the eye are the first synapses in the auditory and visual system. These specialized synapses transmit a large amount of sensory information in a fast and efficient manner. Moreover, both small and large signals with widely variable kinetics must be quickly encoded and reliably transmitted to allow an animal to rapidly monitor and react to its environment. Here we briefly review some aspects of these primary synapses, which are characterized by a synaptic ribbon in their active zones of transmitter release. We propose that these synapses are themselves highly specialized for the task at hand. Photoreceptor and bipolar cell ribbon synapses in the retina appear to have versatile properties that permit both tonic and phasic transmitter release. This allows them to transmit changes of both luminance and contrast within a visual field at different ambient light levels. By contrast, hair cell ribbon synapses are specialized for a highly synchronous form of multivesicular release that may be critical for phase locking to low-frequency sound-evoked signals at both low and high sound intensities. The microarchitecture of a hair cell synapse may be such that the opening of a single Ca(2+) channel evokes the simultaneous exocytosis of multiple synaptic vesicles. Thus, the differing demands of sensory encoding in the eye and ear generate diverse designs and capabilities for their ribbon synapses.
Figures
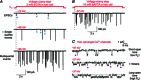
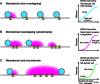
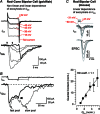

References
-
- Augustine GJ, Adler EM, Charlton MP. The calcium signal for transmitter secretion from presynaptic nerve terminals. Ann N Y Acad Sci. 1991;635:365–381. - PubMed
-
- Beutner D, Voets T, Neher E, Moser T. Calcium dependence of exocytosis and endocytosis at the cochlear inner hair cell afferent synapse. Neuron. 2001;29:681–690. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

