Docetaxel-carboplatin in combination with erlotinib and/or bevacizumab in patients with non-small cell lung cancer
- PMID: 23467839
- PMCID: PMC3589083
- DOI: 10.2147/OTT.S42245
Docetaxel-carboplatin in combination with erlotinib and/or bevacizumab in patients with non-small cell lung cancer
Abstract
Background: Bevacizumab and erlotinib have been demonstrated to prolong overall survival in patients with non-squamous non-small cell lung cancer (NSCLC). We designed a four-arm Phase III trial to evaluate the efficacy and toxicity of the combination of docetaxel, carboplatin, bevacizumab, and erlotinib in the first-line treatment of patients with NSCLC.
Methods: A total of 229 patients with stage IIIb/IV non-squamous NSCLC were treated with two cycles of carboplatin (area under the concentration-time curve 5.5) and docetaxel 100 mg/m2 as chemotherapy. After completion of two treatment cycles, patients were evaluated for response and divided into four groups: 61/229 continued with four more cycles of chemotherapy (control group), 52/229 received chemotherapy plus erlotinib 150 mg daily, 56/229 received chemotherapy plus bevacizumab 7.5 mg/kg, and 60/229 were treated with the combination of chemotherapy, erlotinib, and bevacizumab until disease progression. The primary endpoint was overall survival.
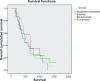
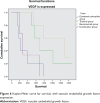
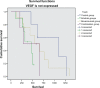
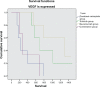
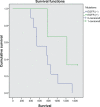
Results: Over 4 years of follow-up, there was no statistically significant difference in survival and time to progression between the four treatment groups. After two cycles of chemotherapy, responders and nonresponders were divided according to their response in order to examine the role of initial response as an independent factor in survival and response when a biological agent is combined with chemotherapy. Nonresponders, who received additional therapy with bevacizumab or combination therapy, had a survival benefit [657 days (95% confidence interval 349-970) and 681 days (95% confidence interval 315-912), respectively], which was statistically significant compared with continuation of cytotoxic chemotherapy (P < 0.001). The combination therapy had a safety profile comparable with that of bevacizumab and erlotinib taken individually.
Conclusion: Administration of bevacizumab and erlotinib in combination with first-line chemotherapy, followed by bevacizumab and erlotinib monotherapy as maintenance, showed promising results in patients with NSCLC, with reduced toxicity as compared with chemotherapy alone, but did not translate into longer overall survival.
Keywords: bevacizumab; epidermal growth factor receptor; erlotinib; non-small cell lung cancer; vascular endothelial growth factor.
Figures
References
-
- Jung YD, Mansfield PF, Akagi M, et al. Effects of combination antivascular endothelial growth factor receptor and anti-epidermal growth factor receptor therapies on the growth of gastric cancer in a nude mouse model. Eur J Cancer. 2002;38(8):1133–1140. - PubMed
-
- Gridelli C, Maione P, Rossi A, De Marinis F. The role of bevacizumab in the treatment of non-small cell lung cancer: current indications and future developments. Oncologist. 2007;12(10):1183–1193. - PubMed
-
- Sandler A, Gray R, Perry MC, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. 2006;355(24):2542–2550. - PubMed
-
- Reck M, von Pawel J, Zatloukal P, et al. Phase III trial of cisplatin plus gemcitabine with either placebo or bevacizumab as first-line therapy for nonsquamous non-small-cell lung cancer: AVAIL. J Clin Oncol. 2009;27(8):1227–1234. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

