Nuclear translocation of cardiac G protein-Coupled Receptor kinase 5 downstream of select Gq-activating hypertrophic ligands is a calmodulin-dependent process
- PMID: 23472081
- PMCID: PMC3589474
- DOI: 10.1371/journal.pone.0057324
Nuclear translocation of cardiac G protein-Coupled Receptor kinase 5 downstream of select Gq-activating hypertrophic ligands is a calmodulin-dependent process
Abstract
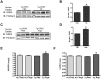
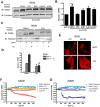
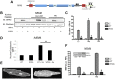
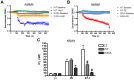
G protein-Coupled Receptors (GPCRs) kinases (GRKs) play a crucial role in regulating cardiac hypertrophy. Recent data from our lab has shown that, following ventricular pressure overload, GRK5, a primary cardiac GRK, facilitates maladaptive myocyte growth via novel nuclear localization. In the nucleus, GRK5's newly discovered kinase activity on histone deacetylase 5 induces hypertrophic gene transcription. The mechanisms governing the nuclear targeting of GRK5 are unknown. We report here that GRK5 nuclear accumulation is dependent on Ca(2+)/calmodulin (CaM) binding to a specific site within the amino terminus of GRK5 and this interaction occurs after selective activation of hypertrophic Gq-coupled receptors. Stimulation of myocytes with phenylephrine or angiotensinII causes GRK5 to leave the sarcolemmal membrane and accumulate in the nucleus, while the endothelin-1 does not cause nuclear GRK5 localization. A mutation within the amino-terminus of GRK5 negating CaM binding attenuates GRK5 movement from the sarcolemma to the nucleus and, importantly, overexpression of this mutant does not facilitate cardiac hypertrophy and related gene transcription in vitro and in vivo. Our data reveal that CaM binding to GRK5 is a physiologically relevant event that is absolutely required for nuclear GRK5 localization downstream of hypertrophic stimuli, thus facilitating GRK5-dependent regulation of maladaptive hypertrophy.
Conflict of interest statement
Figures


References
-
- Hata JA, Koch WJ (2003) Phosphorylation of G protein-coupled receptors: GPCR kinases in heart disease. Mol Interv 3: 264–272. - PubMed
-
- Premont RT, Gainetdinov RR (2007) Physiological roles of G protein-coupled receptor kinases and arrestins. Annu Rev Physiol 69: 511–534. - PubMed
-
- Inglese J, Freedman NJ, Koch WJ, Lefkowitz RJ (1993) Structure and mechanism of the G protein-coupled receptor kinases. J Biol Chem 268: 23735–23738. - PubMed
-
- Metaye T, Gibelin H, Perdrisot R, Kraimps JL (2005) Pathophysiological roles of G-protein-coupled receptor kinases. Cell Signal 17: 917–928. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

