CD11c+ alveolar macrophages are a source of IL-23 during lipopolysaccharide-induced acute lung injury
- PMID: 23481504
- PMCID: PMC3648809
- DOI: 10.1097/SHK.0b013e31828f9c92
CD11c+ alveolar macrophages are a source of IL-23 during lipopolysaccharide-induced acute lung injury
Abstract
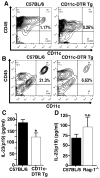
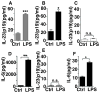
Acute lung injury (ALI) is a severe pulmonary disease causing high numbers of fatalities worldwide. Innate immune responses are an integral part of the pathophysiologic events during ALI. Interleukin 23 (IL-23) is a proinflammatory mediator known to direct the inflammatory responses in various settings of infection, autoimmunity, and cancer. Interleukin 23 has been associated with proliferation and effector functions in T(H)17 cells. Surprisingly, little is known about production of IL-23 during ALI. In this study, we found expression of mRNA for IL-23p19 to be 10-fold elevated in lung homogenates of C57BL/6 mice after lipopolysaccharide (LPS)-induced ALI. Likewise, concentrations of IL-23 significantly increased in bronchoalveolar lavage fluids. Experiments with IL-23-deficient mice showed that endogenous IL-23 was required for production of IL-17A during LPS-ALI. CD11c-diphtheria toxin receptor transgenic mice were used to selectively deplete CD11c cells, the data suggesting that IL-23 production is dependent at least in part on CD11c cells during ALI. No alterations of IL-23 levels were observed in Rag-1-deficient mice as compared with wild-type C57BL/6 mice following ALI. The mouse alveolar macrophage cell line, MH-S, as well as primary alveolar macrophages displayed abundant surface expression of CD11c. Activation of these macrophages by LPS resulted in release of IL-23 in vitro. Our findings identify CD11c macrophages in the lung are likely an important source of IL-23 during ALI, which may be helpful for better understanding of this disease.
Conflict of interest statement
The authors have no commercial or financial conflicts of interests.
Figures



Similar articles
-
Innate-like function of memory Th17 cells for enhancing endotoxin-induced acute lung inflammation through IL-22.Int Immunol. 2016 May;28(5):233-43. doi: 10.1093/intimm/dxv070. Epub 2015 Dec 8. Int Immunol. 2016. PMID: 26647405 Free PMC article.
-
γδ T cells protect against LPS-induced lung injury.J Leukoc Biol. 2016 Feb;99(2):373-86. doi: 10.1189/jlb.4A0115-017RR. Epub 2015 Oct 1. J Leukoc Biol. 2016. PMID: 26428678 Free PMC article.
-
Inhibition of Alveolar Macrophage Pyroptosis Reduces Lipopolysaccharide-induced Acute Lung Injury in Mice.Chin Med J (Engl). 2015 Oct 5;128(19):2638-45. doi: 10.4103/0366-6999.166039. Chin Med J (Engl). 2015. PMID: 26415803 Free PMC article.
-
[Losartan modulates T helper type 1 cells and T helper type 17 cells-mediated responses in a mouse model of lipopolysaccharide-induced acute lung injury].Zhonghua Nei Ke Za Zhi. 2014 Oct;53(10):804-8. Zhonghua Nei Ke Za Zhi. 2014. PMID: 25567153 Chinese.
-
Epigenetic mechanisms of alveolar macrophage activation in chemical-induced acute lung injury.Front Immunol. 2024 Nov 8;15:1488913. doi: 10.3389/fimmu.2024.1488913. eCollection 2024. Front Immunol. 2024. PMID: 39582870 Free PMC article. Review.
Cited by
-
Elevated prostaglandin E2 post-bone marrow transplant mediates interleukin-1β-related lung injury.Mucosal Immunol. 2018 Mar;11(2):319-332. doi: 10.1038/mi.2017.51. Epub 2017 Jun 7. Mucosal Immunol. 2018. PMID: 28589946 Free PMC article.
-
Weathering the Storm: How Age and Biologics Influence the COVID-19 Cytokine Surge.Pathogens. 2025 Apr 3;14(4):346. doi: 10.3390/pathogens14040346. Pathogens. 2025. PMID: 40333142 Free PMC article. Review.
-
Allergic airway inflammation induces upregulation of the expression of IL-23R by macrophages and not in CD3 + T cells and CD11c+F4/80- dendritic cells of the lung.Cell Tissue Res. 2022 Jul;389(1):85-98. doi: 10.1007/s00441-021-03538-0. Epub 2022 Apr 27. Cell Tissue Res. 2022. PMID: 35475923 Free PMC article.
-
A novel chalcone derivative exerts anti-inflammatory and anti-oxidant effects after acute lung injury.Aging (Albany NY). 2019 Sep 24;11(18):7805-7816. doi: 10.18632/aging.102288. Epub 2019 Sep 24. Aging (Albany NY). 2019. PMID: 31553308 Free PMC article.
-
CYP2J2 attenuates metabolic dysfunction in diabetic mice by reducing hepatic inflammation via the PPARγ.Am J Physiol Endocrinol Metab. 2015 Feb 15;308(4):E270-82. doi: 10.1152/ajpendo.00118.2014. Epub 2014 Nov 11. Am J Physiol Endocrinol Metab. 2015. PMID: 25389363 Free PMC article.
References
-
- Goss CH, Brower RG, Hudson LD, Rubenfeld GD. Incidence of acute lung injury in the United States. Critical care medicine. 2003;31:1607–11. - PubMed
-
- Rubenfeld GD, Caldwell E, Peabody E, Weaver J, Martin DP, Neff M, Stern EJ, Hudson LD. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353:1685–93. - PubMed
-
- The Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301–8. - PubMed
-
- Su F, Nguyen ND, Creteur J, Cai Y, Nagy N, Anh-Dung H, Amaral A, Bruzzi de Carvalho F, Chochrad D, Vincent JL. Use of low tidal volume in septic shock may decrease severity of subsequent acute lung injury. Shock. 2004;22:145–50. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

