TCF3, a novel positive regulator of osteogenesis, plays a crucial role in miR-17 modulating the diverse effect of canonical Wnt signaling in different microenvironments
- PMID: 23492770
- PMCID: PMC3613843
- DOI: 10.1038/cddis.2013.65
TCF3, a novel positive regulator of osteogenesis, plays a crucial role in miR-17 modulating the diverse effect of canonical Wnt signaling in different microenvironments
Abstract
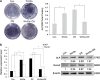
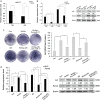
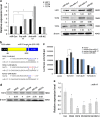
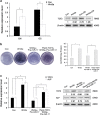
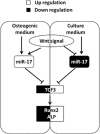
Wnt signaling pathways are a highly conserved pathway, which plays an important role from the embryonic development to bone formation. The effect of Wnt pathway on osteogenesis relies on their cellular environment and the expression of target genes. However, the molecular mechanism of that remains unclear. On the basis of the preliminary results, we observed the contrary effect of canonical Wnt signaling on osteogenic differentiation of periodontal ligament stem cells (PDLSCs) in the different culture environment. Furthermore, we found that the expression level of miR-17 was also varied with the change in the culture environment. Therefore, we hypothesized that miR-17 and canonical Wnt signaling may have potential interactions, particularly the inner regulation relationship in different microenvironments. In this paper, we observed that canonical Wnt signaling promoted osteogenesis of PDLSCs in the fully culture medium, while inhibited it in the osteogenic differentiation medium. Interestingly, alteration in the expression level of endogenous miR-17 could partially reverse the different effect of canonical Wnt signaling. Furthermore, the role of miR-17 was because of its target gene TCF3 (transcription factor 3), a key transcription factor of canonical Wnt pathway. Overexpression of TCF3 attenuated the effect of miR-17 on modulating canonical Wnt signaling. Finally, we elucidated that TCF3 enhanced osteogenesis both in vitro and in vivo. In brief, the different level of miR-17 was the main cause of the different effect of canonical Wnt signaling, and TCF3 was the crucial node of miR-17-canonial Wnt signaling regulation loop. This understanding of microRNAs regulating signaling pathways in different microenvironments may pave the way for fine-tuning the process of osteogenesis in bone-related disorders.
Figures


Similar articles
-
Circular RNA CDR1as regulates osteoblastic differentiation of periodontal ligament stem cells via the miR-7/GDF5/SMAD and p38 MAPK signaling pathway.Stem Cell Res Ther. 2018 Aug 31;9(1):232. doi: 10.1186/s13287-018-0976-0. Stem Cell Res Ther. 2018. PMID: 30170617 Free PMC article.
-
Canonical Wnt signaling differently modulates osteogenic differentiation of mesenchymal stem cells derived from bone marrow and from periodontal ligament under inflammatory conditions.Biochim Biophys Acta. 2014 Mar;1840(3):1125-34. doi: 10.1016/j.bbagen.2013.11.003. Epub 2013 Nov 12. Biochim Biophys Acta. 2014. PMID: 24231680
-
miR-214 promotes periodontal ligament stem cell osteoblastic differentiation by modulating Wnt/β‑catenin signaling.Mol Med Rep. 2017 Dec;16(6):9301-9308. doi: 10.3892/mmr.2017.7821. Epub 2017 Oct 19. Mol Med Rep. 2017. PMID: 29152645 Free PMC article.
-
PPARγ and Wnt Signaling in Adipogenic and Osteogenic Differentiation of Mesenchymal Stem Cells.Curr Stem Cell Res Ther. 2016;11(3):216-25. doi: 10.2174/1574888x10666150519093429. Curr Stem Cell Res Ther. 2016. PMID: 25986621 Review.
-
Regulation of Wnt signaling by non-coding RNAs during osteoblast differentiation.Differentiation. 2022 Nov-Dec;128:57-66. doi: 10.1016/j.diff.2022.10.003. Epub 2022 Oct 15. Differentiation. 2022. PMID: 36370525 Review.
Cited by
-
[Functions of non-coding RNAs in the osteogenic differentiation of human periodontal ligament-derived cells].Hua Xi Kou Qiang Yi Xue Za Zhi. 2020 Jun 1;38(3):330-337. doi: 10.7518/hxkq.2020.03.018. Hua Xi Kou Qiang Yi Xue Za Zhi. 2020. PMID: 32573144 Free PMC article. Review. Chinese.
-
Signature of microRNA expression during osteogenic differentiation of bone marrow MSCs reveals a putative role of miR-335-5p in osteoarthritis.BMC Musculoskelet Disord. 2015 Aug 5;16:182. doi: 10.1186/s12891-015-0652-9. BMC Musculoskelet Disord. 2015. PMID: 26243143 Free PMC article.
-
Non-Coding RNAs Steering the Senescence-Related Progress, Properties, and Application of Mesenchymal Stem Cells.Front Cell Dev Biol. 2021 Mar 19;9:650431. doi: 10.3389/fcell.2021.650431. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33816501 Free PMC article. Review.
-
Bioinformatics Analysis Identified miR-584-5p and Key miRNA-mRNA Networks Involved in the Osteogenic Differentiation of Human Periodontal Ligament Stem Cells.Front Genet. 2021 Sep 27;12:750827. doi: 10.3389/fgene.2021.750827. eCollection 2021. Front Genet. 2021. PMID: 34646313 Free PMC article.
-
Bone remodeling induced by mechanical forces is regulated by miRNAs.Biosci Rep. 2018 Jul 2;38(4):BSR20180448. doi: 10.1042/BSR20180448. Print 2018 Aug 31. Biosci Rep. 2018. PMID: 29844019 Free PMC article. Review.
References
-
- Yang F, Yang D, Tu J, Zheng Q, Cai L, Wang L. Strontium enhances osteogenic differentiation of mesenchymal stem cells and in vivo bone formation by activating Wnt/catenin signaling. Stem Cells. 2011;29:981–991. - PubMed
-
- Bennett CN, Ouyang H, Ma YL, Zeng Q, Gerin I, Sousa KM, et al. Wnt10b increases postnatal bone formation by enhancing osteoblast differentiation. J Bone Miner Res. 2007;22:1924–1932. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

