Sustained virological response on second-line antiretroviral therapy following virological failure in HIV-infected patients in rural South Africa
- PMID: 23505529
- PMCID: PMC3594302
- DOI: 10.1371/journal.pone.0058526
Sustained virological response on second-line antiretroviral therapy following virological failure in HIV-infected patients in rural South Africa
Abstract
Objective: This study aims to describe the virological, immunological and clinical efficacy of protease inhibitor (PI)-based second-line antiretroviral therapy (ART) in rural South Africa.
Methods: An observational cohort study was performed on 210 patients (including 39 children) who initiated PI-based second-line therapy at least 12 months prior to data collection. Biannual clinical, immunological and virological monitoring was performed. Primary endpoints were adequate virological response (plasma HIV-1 RNA<400 copies/ml), full virological suppression (plasma HIV-1 RNA<50 copies/ml) and treatment failure (virological failure (plasma HIV-1 RNA>1000 after initial virological response) or on-going viremia (plasma HIV-1 RNA never<400 copies/ml for more than six months)). Data were analyzed by an on-treatment (OT) and intention-to-treat (ITT) approach. Analyses were primarily performed on the group of patients who switched following first-line virological failure.
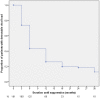
Results: Median duration of follow-up after switch to second-line treatment was 20 months [IQR 11-35]. 191 patients had switched to second-line ART due to first-line virological failure. 139/191 of them (72.8%, ITT) were in care and on treatment at the end of follow-up and 11/191 (5.8%, ITT) had died. After twelve months, an adequate virological response was seen in 92/128 patients (71.9%, OT), of which 78/128 (60.9%, OT) experienced full virological suppression. Virological response remained stable after 24 months. Virological efficacy was similar amongst adult and pediatric patients. As in first-line ART, we observed a lack of correlation between virological failure and WHO-defined immunological failure.
Conclusions: Good virological outcomes following first-line failure can be achieved with PI-based, second-line antiretroviral therapy in both adult and pediatric patients in rural South Africa. Retention rates were high and virological outcomes were sustainable during the two-year follow-up period, although persisting low-level viremia occurred in a subset of patients. The observed viro-immunological dissociation emphasizes the need for virological monitoring.
Conflict of interest statement
Figures

References
-
- (2010) World Health Organization. Antiretroviral therapy for HIV infection in adults and adolescents: Recommendations for a public health approach. 2010 revision. - PubMed
-
- Barth RE, van der Loeff MF, Schuurman R, Hoepelman AI, Wensing AM (2010) Virological follow-up of adult patients in antiretroviral treatment programmes in sub-Saharan Africa: a systematic review. Lancet Infect Dis 10: 155–166. - PubMed
-
- Palombi L, Marazzi MC, Guidotti G, Germano P, Buonomo E, et al. (2009) Incidence and predictors of death, retention, and switch to second-line regimens in antiretroviral- treated patients in sub-Saharan African Sites with comprehensive monitoring availability. Clin Infect Dis 48: 115–122. - PubMed
-
- Zachariah R, Fitzgerald M, Massaquoi M, Pasulani O, Arnould L, et al. (2006) Risk factors for high early mortality in patients on antiretroviral treatment in a rural district of Malawi. Aids 20: 2355–2360. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

