Houttuynia cordata Thunb extract modulates G0/G1 arrest and Fas/CD95-mediated death receptor apoptotic cell death in human lung cancer A549 cells
- PMID: 23506616
- PMCID: PMC3610241
- DOI: 10.1186/1423-0127-20-18
Houttuynia cordata Thunb extract modulates G0/G1 arrest and Fas/CD95-mediated death receptor apoptotic cell death in human lung cancer A549 cells
Abstract
Background: Houttuynia cordata Thunb (HCT) is commonly used in Taiwan and other Asian countries as an anti-inflammatory, antibacterial and antiviral herbal medicine. In this study, we investigated the anti-human lung cancer activity and growth inhibition mechanisms of HCT in human lung cancer A549 cells.
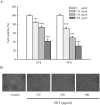
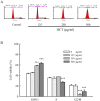
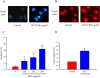
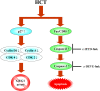
Results: In order to investigate effects of HCT on A549 cells, MTT assay was used to evaluate cell viability. Flow cytometry was employed for cell cycle analysis, DAPI staining, and the Comet assay was used for DNA fragmentation and DNA condensation. Western blot analysis was used to analyze cell cycle and apoptotic related protein levels. HCT induced morphological changes including cell shrinkage and rounding. HCT increased the G0/G1 and Sub-G1 cell (apoptosis) populations and HCT increased DNA fragmentation and DNA condensation as revealed by DAPI staining and the Comet assay. HCT induced activation of caspase-8 and caspase-3. Fas/CD95 protein levels were increased in HCT-treated A549 cells. The G0/G1 phase and apoptotic related protein levels of cyclin D1, cyclin A, CDK 4 and CDK 2 were decreased, and p27, caspase-8 and caspase-3 were increased in A549 cells after HCT treatment.
Conclusions: The results demonstrated that HCT-induced G0/G1 phase arrest and Fas/CD95-dependent apoptotic cell death in A549 cells.
Figures

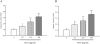
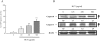
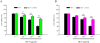
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

