Cyclophilin D deficiency rescues Aβ-impaired PKA/CREB signaling and alleviates synaptic degeneration
- PMID: 23507145
- PMCID: PMC3868643
- DOI: 10.1016/j.bbadis.2013.03.004
Cyclophilin D deficiency rescues Aβ-impaired PKA/CREB signaling and alleviates synaptic degeneration
Abstract
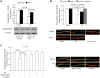
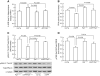
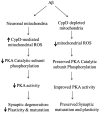
The coexistence of neuronal mitochondrial pathology and synaptic dysfunction is an early pathological feature of Alzheimer's disease (AD). Cyclophilin D (CypD), an integral part of mitochondrial permeability transition pore (mPTP), is involved in amyloid beta (Aβ)-instigated mitochondrial dysfunction. Blockade of CypD prevents Aβ-induced mitochondrial malfunction and the consequent cognitive impairments. Here, we showed the elimination of reactive oxygen species (ROS) by antioxidants probucol or superoxide dismutase (SOD)/catalase blocks Aβ-mediated inactivation of protein kinase A (PKA)/cAMP regulatory-element-binding (CREB) signal transduction pathway and loss of synapse, suggesting the detrimental effects of oxidative stress on neuronal PKA/CREB activity. Notably, neurons lacking CypD significantly attenuate Aβ-induced ROS. Consequently, CypD-deficient neurons are resistant to Aβ-disrupted PKA/CREB signaling by increased PKA activity, phosphorylation of PKA catalytic subunit (PKA C), and CREB. In parallel, lack of CypD protects neurons from Aβ-induced loss of synapses and synaptic dysfunction. Furthermore, compared to the mAPP mice, CypD-deficient mAPP mice reveal less inactivation of PKA-CREB activity and increased synaptic density, attenuate abnormalities in dendritic spine maturation, and improve spontaneous synaptic activity. These findings provide new insights into a mechanism in the crosstalk between the CypD-dependent mitochondrial oxidative stress and signaling cascade, leading to synaptic injury, functioning through the PKA/CREB signal transduction pathway.
Keywords: Alzheimer's disease; Amyloid beta; Mitochondrial permeability transition; Oxidative stress; PKA/CREB signaling; Synaptic alteration.
Copyright © 2013 Elsevier B.V. All rights reserved.
Conflict of interest statement
Figures

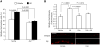



References
-
- von Gunten A, Kovari E, Bussiere T, Rivara CB, Gold G, Bouras C, Hof PR, Giannakopoulos P. Cognitive impact of neuronal pathology in the entorhinal cortex and CA1 field in Alzheimer's disease. Neurobiol Aging. 2006;27:270–277. - PubMed
-
- Cummings BJ, Pike CJ, Shankle R, Cotman CW. Beta-amyloid deposition and other measures of neuropathology predict cognitive status in Alzheimer's disease. Neurobiol Aging. 1996;17:921–933. - PubMed
-
- Scheff SW, Price DA, Schmitt FA, Mufson EJ. Hippocampal synaptic loss in early Alzheimer's disease and mild cognitive impairment. Neurobiol Aging. 2006;27:1372–1384. - PubMed
-
- Selkoe DJ. Alzheimer's disease is a synaptic failure. Science. 2002;298:789–791. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

