Antitumoral potential of Tunisian snake venoms secreted phospholipases A2
- PMID: 23509718
- PMCID: PMC3581298
- DOI: 10.1155/2013/391389
Antitumoral potential of Tunisian snake venoms secreted phospholipases A2
Abstract
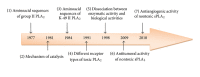
Phospholipases type A2 (PLA2s) are the most abundant proteins found in Viperidae snake venom. They are quite fascinating from both a biological and structural point of view. Despite similarity in their structures and common catalytic properties, they exhibit a wide spectrum of pharmacological activities. Besides being hydrolases, secreted phospholipases A2 (sPLA2) are an important group of toxins, whose action at the molecular level is still a matter of debate. These proteins can display toxic effects by different mechanisms. In addition to neurotoxicity, myotoxicity, hemolytic activity, antibacterial, anticoagulant, and antiplatelet effects, some venom PLA2s show antitumor and antiangiogenic activities by mechanisms independent of their enzymatic activity. This paper aims to discuss original finding against anti-tumor and anti-angiogenic activities of sPLA2 isolated from Tunisian vipers: Cerastes cerastes and Macrovipera lebetina, representing new tools to target specific integrins, mainly, α5β1 and αv integrins.
Figures

Similar articles
-
MVL-PLA2, a phospholipase A2 from Macrovipera lebetina transmediterranea venom, inhibits tumor cells adhesion and migration.Matrix Biol. 2009 May;28(4):188-93. doi: 10.1016/j.matbio.2009.03.007. Epub 2009 Apr 5. Matrix Biol. 2009. PMID: 19351557
-
Functional Characterization and Anti-Tumor Effect of a Novel Group II Secreted Phospholipase A2 from Snake Venom of Saudi Cerastes cerates gasperetti.Molecules. 2023 Sep 8;28(18):6517. doi: 10.3390/molecules28186517. Molecules. 2023. PMID: 37764293 Free PMC article.
-
Snake venomics: comparative analysis of the venom proteomes of the Tunisian snakes Cerastes cerastes, Cerastes vipera and Macrovipera lebetina.Proteomics. 2005 Nov;5(16):4223-35. doi: 10.1002/pmic.200402024. Proteomics. 2005. PMID: 16206329
-
Snake venom phospholipase A2 inhibitors: medicinal chemistry and therapeutic potential.Curr Top Med Chem. 2007;7(8):743-56. doi: 10.2174/156802607780487614. Curr Top Med Chem. 2007. PMID: 17456038 Review.
-
[Snake venom disintegrins: classification and therapeutic potential].Arch Inst Pasteur Tunis. 2007;84(1-4):29-37. Arch Inst Pasteur Tunis. 2007. PMID: 19388581 Review. French.
Cited by
-
Evidence for in vitro antiophidian properties of aqueous buds extract of Eucalyptus against Montivipera bornmuelleri venom.J Venom Res. 2017 Oct 18;8:25-30. eCollection 2017. J Venom Res. 2017. PMID: 29285352 Free PMC article.
-
Antischistosomal Potential of Animal-Derived Natural Products and Compounds.Microorganisms. 2025 Feb 11;13(2):397. doi: 10.3390/microorganisms13020397. Microorganisms. 2025. PMID: 40005763 Free PMC article. Review.
-
Cytotoxic and anticancer properties of the Malaysian mangrove pit viper (Trimeresurus purpureomaculatus) venom and its disintegrin (purpureomaculin).J Venom Anim Toxins Incl Trop Dis. 2020 Jul 17;26:e20200013. doi: 10.1590/1678-9199-JVATITD-2020-0013. eCollection 2020. J Venom Anim Toxins Incl Trop Dis. 2020. PMID: 32742279 Free PMC article.
-
Conotoxin Prediction: New Features to Increase Prediction Accuracy.Toxins (Basel). 2023 Nov 3;15(11):641. doi: 10.3390/toxins15110641. Toxins (Basel). 2023. PMID: 37999504 Free PMC article.
-
A study of ribonuclease activity in venom of vietnam cobra.J Anim Sci Technol. 2017 Sep 25;59:20. doi: 10.1186/s40781-017-0145-5. eCollection 2017. J Anim Sci Technol. 2017. PMID: 29021904 Free PMC article.
References
-
- El-Refaei MF, Sarkar NH. Snake venom inhibits the growth of mouse mammary tumor cells in vitro and in vivo. Toxicon. 2009;54:33–41. - PubMed
-
- Guimarães-Gomes V, Oliveira-Carvalho AL, L. M. Junqueira-De-Azevedo ID, et al. Cloning, characterization, and structural analysis of a C-type lectin from Bothrops insularis (BiL) venom. Archives of Biochemistry and Biophysics. 2004;432(1):1–11. - PubMed
-
- Stocker K. Use of snake venom proteins in medicine. Schweizerische Medizinische Wochenschrift. 1999;129(6):205–216. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

