Enterocyte loss of polarity and gut wound healing rely upon the F-actin-severing function of villin
- PMID: 23520048
- PMCID: PMC3625339
- DOI: 10.1073/pnas.1218446110
Enterocyte loss of polarity and gut wound healing rely upon the F-actin-severing function of villin
Abstract
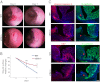
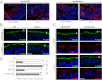
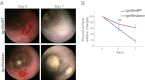
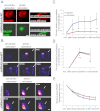
Efficient wound healing is required to maintain the integrity of the intestinal epithelial barrier because of its constant exposure to a large variety of environmental stresses. This process implies a partial cell depolarization and the acquisition of a motile phenotype that involves rearrangements of the actin cytoskeleton. Here we address how polarized enterocytes harboring actin-rich apical microvilli undergo extensive cell remodeling to drive injury repair. Using live imaging technologies, we demonstrate that enterocytes in vitro and in vivo rapidly depolarize their microvilli at the wound edge. Through its F-actin-severing activity, the microvillar actin-binding protein villin drives both apical microvilli disassembly in vitro and in vivo and promotes lamellipodial extension. Photoactivation experiments indicate that microvillar actin is mobilized at the lamellipodium, allowing optimal migration. Finally, efficient repair of colonic mechanical injuries requires villin severing of F-actin, emphasizing the importance of villin function in intestinal homeostasis. Thus, villin severs F-actin to ensure microvillus depolarization and enterocyte remodeling upon injury. This work highlights the importance of specialized apical pole disassembly for the repolarization of epithelial cells initiating migration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Villin enhances hepatocyte growth factor-induced actin cytoskeleton remodeling in epithelial cells.Mol Biol Cell. 2003 Nov;14(11):4641-53. doi: 10.1091/mbc.e03-02-0091. Epub 2003 Aug 22. Mol Biol Cell. 2003. PMID: 12937273 Free PMC article.
-
Villin-induced growth of microvilli is reversibly inhibited by cytochalasin D.J Cell Sci. 1993 Jul;105 ( Pt 3):765-75. doi: 10.1242/jcs.105.3.765. J Cell Sci. 1993. PMID: 8408303
-
In vivo, villin is required for Ca(2+)-dependent F-actin disruption in intestinal brush borders.J Cell Biol. 1999 Aug 23;146(4):819-30. doi: 10.1083/jcb.146.4.819. J Cell Biol. 1999. PMID: 10459016 Free PMC article.
-
From the structure to the function of villin, an actin-binding protein of the brush border.Bioessays. 1990 Sep;12(9):403-8. doi: 10.1002/bies.950120902. Bioessays. 1990. PMID: 2256904 Review.
-
Regulation of cell structure and function by actin-binding proteins: villin's perspective.FEBS Lett. 2008 Jun 18;582(14):2128-39. doi: 10.1016/j.febslet.2008.02.040. Epub 2008 Feb 26. FEBS Lett. 2008. PMID: 18307996 Free PMC article. Review.
Cited by
-
The F-actin modifier villin regulates insulin granule dynamics and exocytosis downstream of islet cell autoantigen 512.Mol Metab. 2016 Jun 10;5(8):656-668. doi: 10.1016/j.molmet.2016.05.015. eCollection 2016 Aug. Mol Metab. 2016. PMID: 27656403 Free PMC article.
-
Taste cell-expressed α-glucosidase enzymes contribute to gustatory responses to disaccharides.Proc Natl Acad Sci U S A. 2016 May 24;113(21):6035-40. doi: 10.1073/pnas.1520843113. Epub 2016 May 9. Proc Natl Acad Sci U S A. 2016. PMID: 27162343 Free PMC article.
-
Manipulation of microvillar proteins during Salmonella enterica invasion results in brush border effacement and actin remodeling.Front Cell Infect Microbiol. 2023 Mar 2;13:1137062. doi: 10.3389/fcimb.2023.1137062. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 36936760 Free PMC article.
-
Gut homeostasis, injury, and healing: New therapeutic targets.World J Gastroenterol. 2022 May 7;28(17):1725-1750. doi: 10.3748/wjg.v28.i17.1725. World J Gastroenterol. 2022. PMID: 35633906 Free PMC article. Review.
-
By moonlighting in the nucleus, villin regulates epithelial plasticity.Mol Biol Cell. 2016 Feb 1;27(3):535-48. doi: 10.1091/mbc.E15-06-0453. Epub 2015 Dec 10. Mol Biol Cell. 2016. PMID: 26658611 Free PMC article.
References
-
- Blikslager AT, Moeser AJ, Gookin JL, Jones SL, Odle J. Restoration of barrier function in injured intestinal mucosa. Physiol Rev. 2007;87(2):545–564. - PubMed
-
- Fenteany G, Janmey PA, Stossel TP. Signaling pathways and cell mechanics involved in wound closure by epithelial cell sheets. Curr Biol. 2000;10(14):831–838. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

