Characterization of transgenic Gfrp knock-in mice: implications for tetrahydrobiopterin in modulation of normal tissue radiation responses
- PMID: 23521531
- PMCID: PMC3936502
- DOI: 10.1089/ars.2012.5025
Characterization of transgenic Gfrp knock-in mice: implications for tetrahydrobiopterin in modulation of normal tissue radiation responses
Abstract
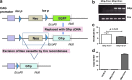
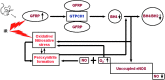
Aims: The free radical scavenger and nitric oxide synthase cofactor, 5,6,7,8-tetrahydrobiopterin (BH4), plays a well-documented role in many disorders associated with oxidative stress, including normal tissue radiation responses. Radiation exposure is associated with decreased BH4 levels, while BH4 supplementation attenuates aspects of radiation toxicity. The endogenous synthesis of BH4 is catalyzed by the enzyme guanosine triphosphate cyclohydrolase I (GTPCH1), which is regulated by the inhibitory GTP cyclohydrolase I feedback regulatory protein (GFRP). We here report and characterize a novel, Cre-Lox-driven, transgenic mouse model that overexpresses Gfrp.
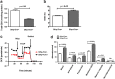
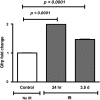
Results: Compared to control littermates, transgenic mice exhibited high transgene copy numbers, increased Gfrp mRNA and GFRP expression, enhanced GFRP-GTPCH1 interaction, reduced BH4 levels, and low glutathione (GSH) levels and differential mitochondrial bioenergetic profiles. After exposure to total body irradiation, transgenic mice showed decreased BH4/7,8-dihydrobiopterin ratios, increased vascular oxidative stress, and reduced white blood cell counts compared with controls.
Innovation and conclusion: This novel Gfrp knock-in transgenic mouse model allows elucidation of the role of GFRP in the regulation of BH4 biosynthesis. This model is a valuable tool to study the involvement of BH4 in whole body and tissue-specific radiation responses and other conditions associated with oxidative stress.
Figures



Similar articles
-
Reduction of radiation-induced vascular nitrosative stress by the vitamin E analog γ-tocotrienol: evidence of a role for tetrahydrobiopterin.Int J Radiat Oncol Biol Phys. 2011 Mar 1;79(3):884-91. doi: 10.1016/j.ijrobp.2010.08.032. Epub 2010 Oct 13. Int J Radiat Oncol Biol Phys. 2011. PMID: 20950957 Free PMC article.
-
Changes in tetrahydrobiopterin levels in endothelial cells and adult cardiomyocytes induced by LPS and hydrogen peroxide--a role for GFRP?Free Radic Biol Med. 2005 Feb 15;38(4):481-91. doi: 10.1016/j.freeradbiomed.2004.11.004. Free Radic Biol Med. 2005. PMID: 15649650
-
Ligand binding to the inhibitory and stimulatory GTP cyclohydrolase I/GTP cyclohydrolase I feedback regulatory protein complexes.Protein Sci. 2001 Apr;10(4):871-8. doi: 10.1110/ps.38501. Protein Sci. 2001. PMID: 11274478 Free PMC article.
-
Maintenance of cellular tetrahydrobiopterin homeostasis.BMB Rep. 2010 Sep;43(9):584-92. doi: 10.5483/BMBRep.2010.43.9.584. BMB Rep. 2010. PMID: 20846489 Review.
-
Tetrahydrobiopterin in energy metabolism and metabolic diseases.Pharmacol Res. 2020 Jul;157:104827. doi: 10.1016/j.phrs.2020.104827. Epub 2020 Apr 26. Pharmacol Res. 2020. PMID: 32348841 Review.
Cited by
-
Inhibition of Ferroptosis by Mesenchymal Stem Cell-Derived Exosomes in Acute Spinal Cord Injury: Role of Nrf2/GCH1/BH4 Axis.Neurospine. 2024 Jun;21(2):642-655. doi: 10.14245/ns.2448038.019. Epub 2024 Jun 30. Neurospine. 2024. PMID: 38955534 Free PMC article.
-
In Vitro Toxicity and Epigenotoxicity of Different Types of Ambient Particulate Matter.Toxicol Sci. 2015 Dec;148(2):473-87. doi: 10.1093/toxsci/kfv200. Epub 2015 Sep 4. Toxicol Sci. 2015. PMID: 26342214 Free PMC article.
-
The Critical Role of Tetrahydrobiopterin (BH4) Metabolism in Modulating Radiosensitivity: BH4/NOS Axis as an Angel or a Devil.Front Oncol. 2021 Aug 27;11:720632. doi: 10.3389/fonc.2021.720632. eCollection 2021. Front Oncol. 2021. PMID: 34513700 Free PMC article. Review.
-
Modulation of Radiation Response by the Tetrahydrobiopterin Pathway.Antioxidants (Basel). 2015 Jan 22;4(1):68-81. doi: 10.3390/antiox4010068. Antioxidants (Basel). 2015. PMID: 26785338 Free PMC article. Review.
-
Tocotrienol-Rich Fraction from Rice Bran Demonstrates Potent Radiation Protection Activity.Evid Based Complement Alternat Med. 2015;2015:148791. doi: 10.1155/2015/148791. Epub 2015 Aug 26. Evid Based Complement Alternat Med. 2015. PMID: 26425129 Free PMC article.
References
-
- Alegria-Schaffer A, Lodge A, and Vattem K. Performing and optimizing Western blots with an emphasis on chemiluminescent detection. Methods Enzymol 463: 573–599, 2009 - PubMed
-
- Bauersachs J, and Widder JD. Tetrahydrobiopterin, endothelial nitric oxide synthase, and mitochondrial function in the heart. Hypertension 53: 907–908, 2009 - PubMed
-
- Brown GC. Nitric oxide regulates mitochondrial respiration and cell functions by inhibiting cytochrome oxidase. FEBS Lett 369: 136–139, 1995 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

