Eye-mediated induction of specific immune tolerance to encephalitogenic antigens
- PMID: 23522052
- PMCID: PMC6493606
- DOI: 10.1111/cns.12087
Eye-mediated induction of specific immune tolerance to encephalitogenic antigens
Abstract
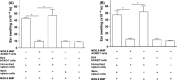
Aims: Administration of antigens into the anterior chamber (AC) of the eye induces a form of antigen-specific immune tolerance termed anterior chamber-associated immune deviation (ACAID). This immune tolerance effectively impairs host delayed-type hypersensitivity (DTH) responses. We hypothesized that ACAID could be generated in BALB/c mice following AC inoculation of the encephalitogenic antigens myelin oligodendrocyte glycoprotein (MOG) and myelin basic protein (MBP).
Methods: We used DTH assays and local adoptive transfer (LAT) assays to test whether MOG/MBP-induced ACAID following their administration into the AC, whether they elicited this immune tolerance via CD8(+) T cells, and whether their AC coadministration (MOG/MBP) induced specific immune tolerance to one or both antigens.
Results: We showed that MOG/MBP-induced AC-mediated specific immune tolerance, as evident from impaired DTH responses. This antigen-driven DTH suppression was solely mediated via splenic CD8(+) T cells as confirmed by LAT assays. Finally, a single AC injection with both antigens was sufficient to induce specific immune tolerance to these antigens, as evident from DTH and LAT assays.
Conclusion: ACAID T-cell regulation could be used as a therapeutic tool in the treatment of complicated autoimmune diseases that involve multiple antigens such as multiple sclerosis.
© 2013 John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Medawar PB. A second study of the behaviour and fate of skin homografts in rabbits: a report to the war wounds committee of the medical research council. J Anat 1945;79:157–176, 154. - PubMed
-
- Forrester JV, Xu H, Lambe T, Cornall R. Immune privilege or privileged immunity? Mucosal Immunol 2008;1:372–381. - PubMed
-
- Ashour HM, Niederkorn JY. Gammadelta t cells promote anterior chamber‐associated immune deviation and immune privilege through their production of il‐10. J Immunol 2006;177:8331–8337. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

