Lymphotoxin signalling in immune homeostasis and the control of microorganisms
- PMID: 23524463
- PMCID: PMC3900493
- DOI: 10.1038/nri3406
Lymphotoxin signalling in immune homeostasis and the control of microorganisms
Abstract
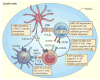
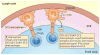
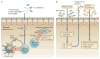
Lymphotoxin (LT) is a member of the tumour necrosis factor (TNF) superfamily that was originally thought to be functionally redundant to TNF, but these proteins were later found to have independent roles in driving lymphoid organogenesis. More recently, LT-mediated signalling has been shown to actively contribute to effector immune responses. LT regulates dendritic cell and CD4(+) T cell homeostasis in the steady state and determines the functions of these cells during pathogenic challenges. The LT receptor pathway is essential for controlling pathogens and even contributes to the regulation of the intestinal microbiota, with recent data suggesting that LT-induced changes in the microbiota promote metabolic disease. In this Review, we discuss these newly defined roles for LT, with a particular focus on how the LT receptor pathway regulates innate and adaptive immune responses to microorganisms.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

Similar articles
-
Lymphotoxin in physiology of lymphoid tissues - Implication for antiviral defense.Cytokine. 2018 Jan;101:39-47. doi: 10.1016/j.cyto.2016.08.018. Epub 2016 Sep 9. Cytokine. 2018. PMID: 27623349 Free PMC article. Review.
-
Lymphotoxin organizes contributions to host defense and metabolic illness from innate lymphoid cells.Cytokine Growth Factor Rev. 2014 Apr;25(2):227-33. doi: 10.1016/j.cytogfr.2013.12.007. Epub 2013 Dec 24. Cytokine Growth Factor Rev. 2014. PMID: 24411493 Free PMC article. Review.
-
ILC3s control splenic cDC homeostasis via lymphotoxin signaling.J Exp Med. 2021 May 3;218(5):e20190835. doi: 10.1084/jem.20190835. J Exp Med. 2021. PMID: 33724364 Free PMC article.
-
Lymphotoxin controls the IL-22 protection pathway in gut innate lymphoid cells during mucosal pathogen challenge.Cell Host Microbe. 2011 Jul 21;10(1):44-53. doi: 10.1016/j.chom.2011.06.002. Cell Host Microbe. 2011. PMID: 21767811 Free PMC article.
-
The Lymphotoxin Network: orchestrating a type I interferon response to optimize adaptive immunity.Cytokine Growth Factor Rev. 2014 Apr;25(2):139-45. doi: 10.1016/j.cytogfr.2014.02.002. Epub 2014 Mar 12. Cytokine Growth Factor Rev. 2014. PMID: 24698108 Free PMC article. Review.
Cited by
-
Cytokines IL-17 and IL-22 in the host response to infection.Pathog Dis. 2016 Dec;74(9):ftw111. doi: 10.1093/femspd/ftw111. Epub 2016 Dec 2. Pathog Dis. 2016. PMID: 27915228 Free PMC article. Review.
-
A human model of asthma exacerbation reveals transcriptional programs and cell circuits specific to allergic asthma.Sci Immunol. 2023 May 12;8(83):eabq6352. doi: 10.1126/sciimmunol.abq6352. Epub 2023 May 5. Sci Immunol. 2023. PMID: 37146132 Free PMC article.
-
Comparative Transcriptomic Analysis Identifies a Range of Immunologically Related Functional Elaborations of Lymph Node Associated Lymphatic and Blood Endothelial Cells.Front Immunol. 2019 Apr 16;10:816. doi: 10.3389/fimmu.2019.00816. eCollection 2019. Front Immunol. 2019. PMID: 31057546 Free PMC article.
-
Associations of lymphotoxin-a (LTA) rs909253 A/G gene polymorphism, plasma level and risk of ankylosing spondylitis in a Chinese Han population.Sci Rep. 2020 Jan 29;10(1):1412. doi: 10.1038/s41598-020-57927-6. Sci Rep. 2020. PMID: 31996699 Free PMC article.
-
Neuroimmune interactions: The bridge between inflammatory bowel disease and the gut microbiota.Clin Transl Med. 2025 May;15(5):e70329. doi: 10.1002/ctm2.70329. Clin Transl Med. 2025. PMID: 40400119 Free PMC article. Review.
References
-
- Eberl G. A new vision of immunity: homeostasis of the superorganism. Mucosal Immunol. 2010;3:450–460. - PubMed
-
- Ware CF, VanArsdale TL, Crowe PD, Browning JL. The ligands and receptors of the lymphotoxin system. Curr Top Microbiol Immunol. 1995;198:175–218. - PubMed
-
- Geuking MB, et al. Intestinal bacterial colonization induces mutualistic regulatory T cell responses. Immunity. 2011;34:794–806. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

